
New Review of Artificial Sweeteners
A new review of research finds a modest but inconsistent benefit from consuming artificial sweeteners over sugar. Their conclusion of a possible backfire effect, however, does not seem to be supported by the studies they review.
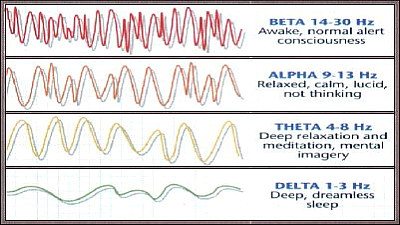
Brain Wave Pseudoscience
iSynchrony has put together a plausible-sounding bit of bafflegab to justify what it sells. The reality of neurology is against their claims.

Amish Farmer Jailed for Selling Snakeoil
An Amish farmer is convicted of selling a caustic poison as patent medicine (and of witness tampering) and yet is defended by "alternative medicine" proponents who apparently want the freedom to be defrauded and harmed.

The Skinny on Saturated Fat
The bottom line is that there are trade-offs and complexities when it comes to biology. A high carbohydrate diet is a risk factor for many diseases, and this is highly genetically variable. Types of sugar matter, weight matters, exercise matters, and genetics matter. The same is true of fat.

Coconut Oil Warning
Coconut oil is promoted as a health food, but a recent warning from the American Heart Association warns that coconut oil is very high in saturated fats and increases your risk for heart disease.
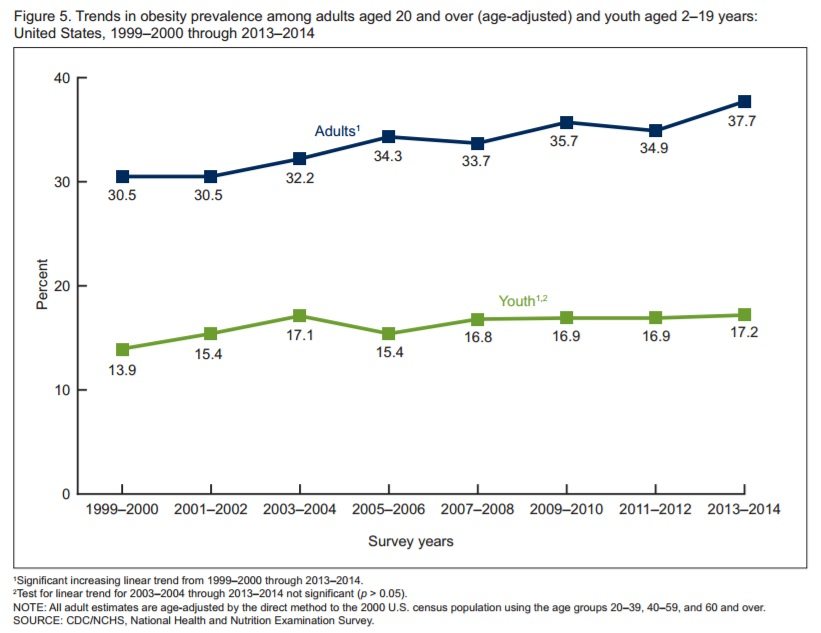
BMI and the Obesity Epidemic
Obesity is a serious and growing health problem worldwide. Ditching BMI as a measure of obesity is not the solution.
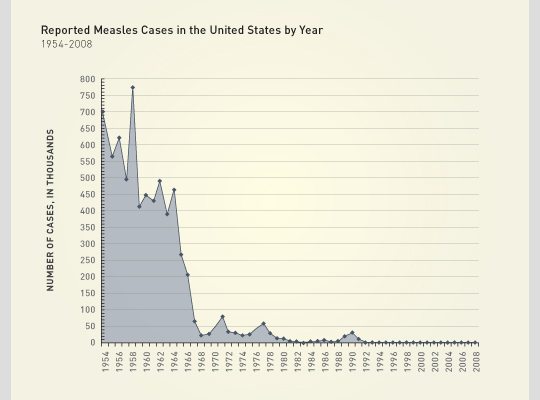
Minnesota Measles
There is currently an outbreak of measles in the Somali population of Minnesota – an outbreak that can be directly linked to antivaccine propaganda.

The Natural Cancer Cure Narrative
Julie Reason and her husband are producing a documentary about her cancer, and efforts to cure it. Based on their comments, they are drawing upon an established and false narrative about the causes and cures of cancer, one that can be dangerous to her, and all other cancer patients.

Another ADHD Denier
John Rosemond, a self-help columnist, denies the science of ADHD with the usual invalid and outdated arguments.


Mast Cell Activation Disorder – Yes, It’s Real
Mast Cell Activation Disorder is real, but there are a large number of fake diagnoses out there. How do you tell the difference?