Category: Herbs & Supplements
Natural Health Products: Loosely regulated, little evidence of benefit, and an industry intent on preserving the status quo
This week’s post will revisit a topic I recently covered, but it’s time-sensitive and needs your input. Health Canada, the Canadian equivalent to the US Food and Drugs Administration, is considering revisions to the way in which it regulates dietary supplements, which are called “natural health products” in Canada. It is rare that a regulator acknowledges that a regulatory system isn’t working,...

Is there a distinct standard of care for “integrative” physicians? The Woliner case
We at SBM argue that there should be a single, science-based standard of care in medicine. Unfortunately, with the rise of "complementary and alternative medicine" (CAM) also called "integrative medicine," there is a separate standard emerging that allows CAM practitioners to get away with using unproven and disproven treatments. The case of Dr. Kenneth Woliner illustrates this problem.
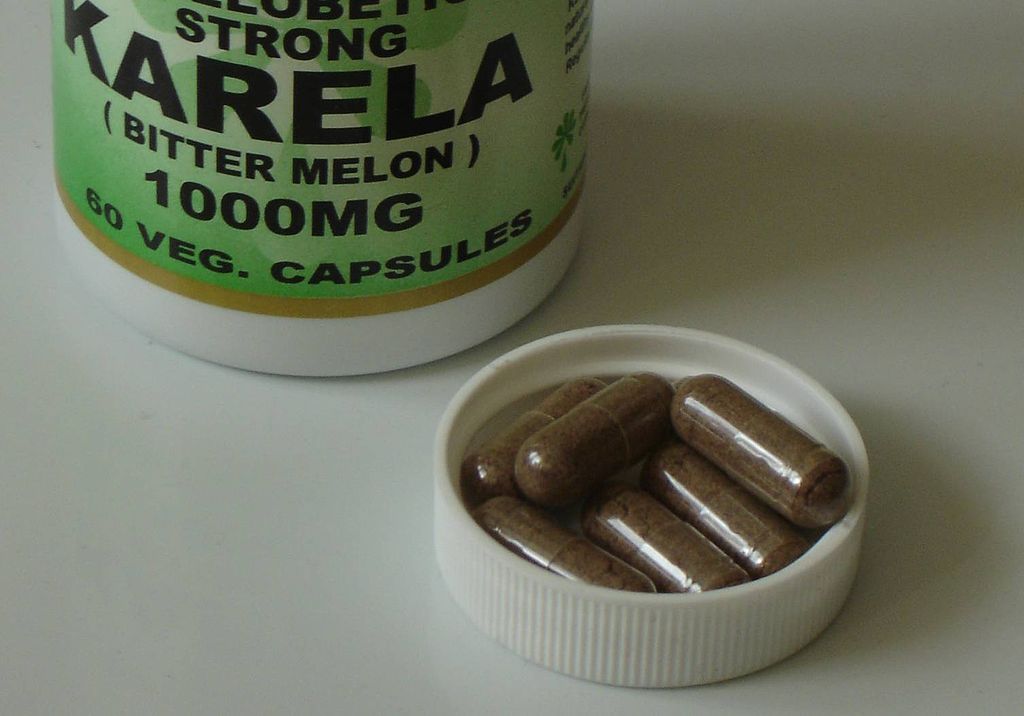
Plavinol and Other Natural Remedies for Diabetes: “Condimentary Medicine”?
We don’t yet have a cure for diabetes, but we have insulin; it controls the disease and allows Type 1 diabetics to lead a relatively normal life instead of suffering and quickly dying as they all did in the pre-insulin era. We know to counsel Type 2 diabetics about weight loss, diet, and exercise; and when those measures are not enough, we...

Fixing the supplement market for consumers
When it comes to regulating and selling dietary supplements, should consumer interests be higher priority than those of manufacturers? While regulations are seemingly created to protect consumers, governments around the world have consistently given manufacturers the upper hand, prioritizing a company’s desire to sell a product over a consumer’s right to a marketplace with safe, effective products. Nowhere is this more the...
“Glyconutrients,” Mannatech, and Ambrotose: Marketing, Not Science
It has been a long time since I first became aware of Mannatech, the multilevel marketing company that sells “glyconutrient” dietary supplements. After its claims were debunked and it lost a court case, it had dropped off my radar; but last month it came roaring back in the form of an email from a reader in South Africa. He said his in-laws...
Oxygen water? You can’t breathe through your stomach
My exercise of choice is running. Despite the heat I’ve been having a great summer, training for the Chicago marathon. I’ve followed the training schedule fanatically since June. But it all came crashing down in one run last week when I moved from the ranks of “marathoner in training” to “injured runner”. With the sudden onset of very sharp, radiating back pain,...
Kratom: another dangerous “natural” remedy
Kratom (Mitragyna speciose) is a tropical tree from Southeast Asia whose leaves are traditionally chewed or prepared as a powder. Native populations chew the leaves to reduce fatigue when doing manual labor, such as working on rubber plantations. It is also used in cultural performances and consumed as a drink prepared from kratom powder. When the Second World War caused an increase...
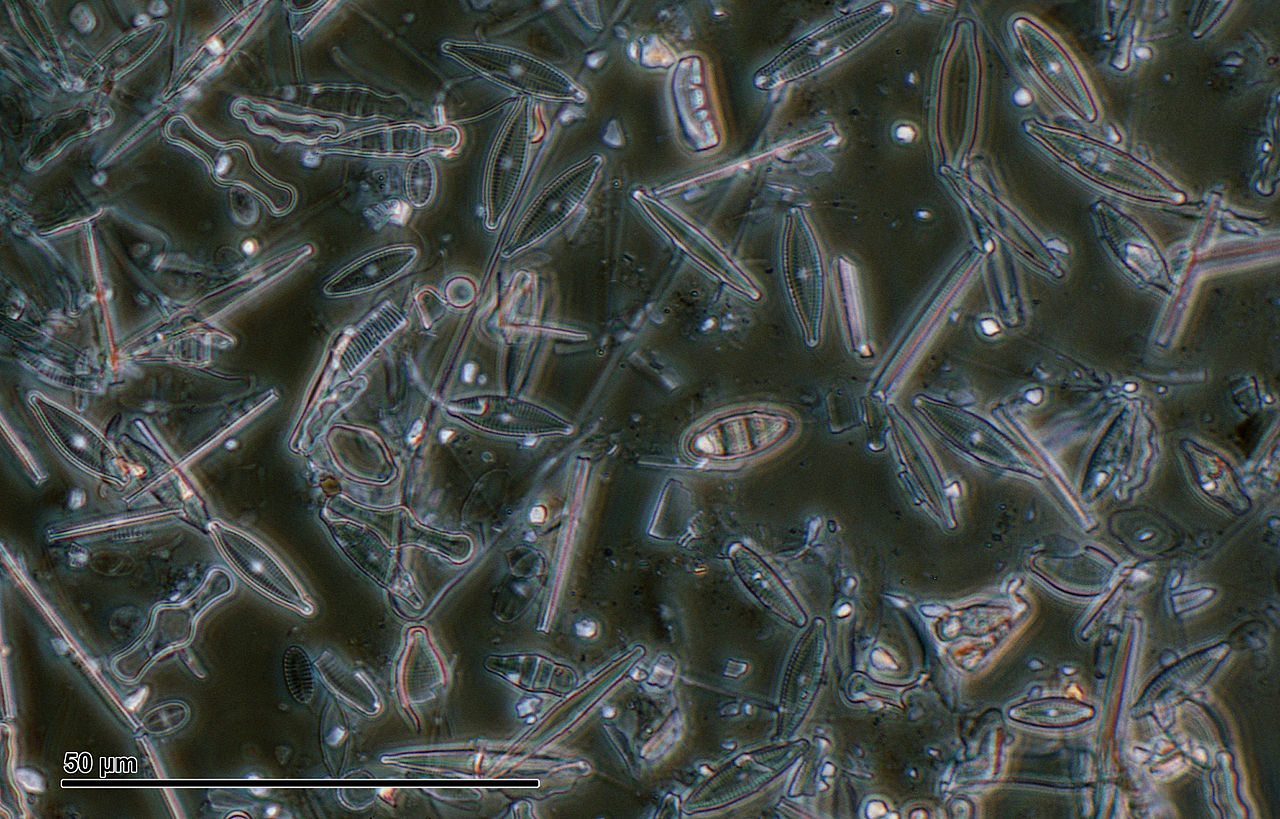
Diatomaceous Earth? No Thank You!
Diatoms are unicellular algae, one of the two major classes of the phytoplankton that constitute the bottom of the food chain in oceans and freshwater. Diatomaceous earth is a soft, siliceous sedimentary rock containing the fossilized skeletal remains of diatoms. It has been used as a bug killer: it is hypothesized that the sharp particles physically cut up the insects and also...

FDA efforts to improve compounded drug safety upsets naturopaths
Favorite naturopathic treatments comprise pumping patients full of dubious mixtures by injection, including IV drips. Naturopaths also employ topicals (salves, ointments and creams), rectal, and vaginal suppositories, and oral medications, such as bio-identical hormone replacement therapy, all made from “natural” substances. According to the American Association of Naturopathic Physicians (AANP) these nutritional, herbal and homeopathic remedies are compounded to meet unique patient...

The THRIVE Experience: Masterful Marketing, Short on Substance
My daughter told me about the latest health fad among her group of acquaintances. She knows people who are spending $300 a month on the THRIVE program and claiming miraculous results. With a skeptic for a mother, my daughter knew enough to question the claims and do her own research; she was not impressed. She concluded that THRIVE was essentially selling caffeine...

