Category: Legal

FDA efforts to improve compounded drug safety upsets naturopaths
Favorite naturopathic treatments comprise pumping patients full of dubious mixtures by injection, including IV drips. Naturopaths also employ topicals (salves, ointments and creams), rectal, and vaginal suppositories, and oral medications, such as bio-identical hormone replacement therapy, all made from “natural” substances. According to the American Association of Naturopathic Physicians (AANP) these nutritional, herbal and homeopathic remedies are compounded to meet unique patient...
Supplements, Lies, and a Lengthy Transcript
On October 21, 1993, there was a hearing before the U.S. Senate Committee for Labor and Human Resources, with the long-winded title: Examining How the Federal Government Should Regulate the Marketing and Use of Dietary Supplements and Related Measures, Including S. 784, To Strengthen Federal Standards with Respect To Dietary Supplements. S. 784, sponsored by Sen. Orrin Hatch, would eventually be enacted...
Change.org Petition: “Naturopaths are not physicians: stop legitimizing pseudoscience”
Britt Hermes, a graduate of the naturopathic college at the alternative medicine-focused Bastyr University, renounced her practice as a naturopathic doctor when she could no longer tolerate the pseudoscience and patient harm that characterizes naturopathy. On this blog and her own, Naturopathic Diaries, she has chronicled the insufficient education and training students receive before being allowed to practice as naturopathic doctors, deficiencies...
Legislative Alchemy: Michigan House Bill 4531 gives naturopaths a broad scope of practice
Michigan House Bill 4531, if passed, would give naturopaths one of the broadest scopes of practice in the U.S., essentially equaling that of a family practice MD or DO. The bill made it through all the necessary House committees and is now before the House for an initial vote determining whether it will proceed further in that body. If it passes there,...
What (if anything) does “natural” mean?
What does the term “natural” mean on a label? Does it mean anything? Should it mean anything? Good questions. And complicated ones, judging from the list of questions the FDA needs your help in answering. The FDA has resisted defining “natural” in food product labeling, including whether foods that are genetically engineered, or contain genetically engineered ingredients, can use the term. Back...
Regulating CAM Aussie Style
CAM proponents view National Health Interview Surveys recording the supposed popularity of CAM, an amorphous conflation of anything from conventional medical advice to mythical methods, as an invitation to unleash even more unproven remedies on the public. My interpretation is quite different. I see the same figures as proof that we are doing too little to protect the public from pseudoscience. In...
Patient Beware: Off-label drug promotion by pharmaceutical companies
Pharmaceutical companies and their sales reps can distribute information, such as medical journal articles, about unapproved (“off-label“) uses of their drugs as long as they adhere to FDA guidelines. However, the FDA takes the position that this information must be distributed separately from information that is “promotional in nature,” i.e., for marketing purposes, a position that is now open to question. Off-label...
Health Savings Accounts: a tax-sheltered way to pay for quackery
If you want money to pay for pseudoscience, but your pesky health insurance company is getting in the way, a Health Savings Account might be just the solution. And if the Health Savings Act of 2016, sponsored by the Big Supplement’s own Senator Orrin Hatch, becomes law, your opportunities will be greatly expanded. First, let’s take a look at Health Savings Accounts...
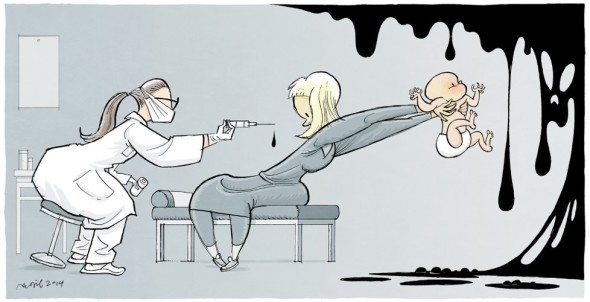
Laws Limiting Vaccine Exemptions Work
It’s nice when a question can be resolved with objective numbers of unequivocal outcomes. Subjective outcomes give scientists a headache. In this case we are talking about the effect of vaccine exemption laws on vaccine compliance rates. The question here is not the ethical one, the rights of parents to determine the fate of their children vs the right of the state...
Docs v. Glocks: government regulation of physician speech
A few years ago, an Ocala, Florida, pediatrician, as part of a routine visit, asked a patient’s mother whether she kept firearms in the home. She refused to answer, feeling the question constituted an invasion of her right to privacy. The pediatrician then terminated the relationship and told the mother she had 30 days to find a new doctor for her child....

