Category: Medical Ethics
Placebo Therapies: Are They Ethical?
Is it ethical to overstate the efficacy of a treatment option, if it might lead to a patient’s enhanced experience of that treatment? Your response to this question may reveal the degree to which you favor Complementary and Alternative Medicine (CAM). Let me explain. As far as I can tell, no CAM treatment has been proven effective beyond placebo. (If you’re not...
Lies, Damned Lies, and ‘Integrative Medicine’
Last week, two events took place in Washington that ought to inspire trepidation in the minds of all who value ethical, rational, science-based medicine and ethical, rational, biomedical research. One was the Senate Panel titled Integrative Care: A Pathway to a Healthier Nation, previously discussed by my fellow bloggers David Gorski, Peter Lipson, and Steve Novella, and also by the indefatigable Orac (here and...
Colorado is Nearer to Promoting Naturopathic Pseudomedicine—Aided by the Colorado Medical Society
This week we’ll take a break from lambasting the National Center for Complementary and Alternative Medicine, as worthy as that task is, in order to confront some of the latest events involving the pseudomedical cult that calls itself “naturopathic medicine.”* Intrepid nurse and anti-healthfraud activist Linda Rosa reports that Colorado is dangerously close to becoming the next state to endorse “NDs” as health...
Obama and Stem Cells
In 2001 George Bush signed an executive order banning federal funding for embryonic stem cell research, except for those lines that were already established. As a result such research ground to a halt in the US. While the order was presented as a compromise, the effect was chilling in its application. No researcher receiving federal dollars (even for a separate project) could...
Yes We Can! We Can Abolish the NCCAM! Part III
A Reminder… …of why we keep harping on this. A couple of days ago The Scientist reported that the “economic stimulus package” may include a windfall for the NIH: Senate OKs big NIH bump Posted by Bob Grant [Entry posted at 4th February 2009 04:12 PM GMT] The US Senate, which is furiously debating the details of the economic stimulus package making its...
Yes We Can! We Can Abolish the NCCAM!
…and in so doing, President Obama, you and we would abolish the NIH’s second most prodigious squanderer of precious research funds! Surprise: The National Cancer Institute (NCI) spends slightly more on humbug than does the Center created for that purpose. All told, the NIH squanders almost 1/3 of a billion dollars per year promoting pseudoscience. I’ve decided to add my two cents to the recent groundswell of demand...
Guest Book Review of “Complementary and Alternative Medicine: Ethics, the Patient, and the Physician”
The following book review was written not by your poster (although I’ve added the hyperlinks), but by his friend Cees Renckens, who is a gynecologist in the Netherlands and the chairman of the Dutch Society against Quackery. A short bio of Dr. Renckens, including references to several articles in English, follows the review. Most impressive to me is that he is, as far as I...
The Weekly Waluation of the Weasel Words of Woo #9
Resurrection! Can y’believe it? The W^5/2™, that cesspool of Afflicted Sophistry and Festering Fallacy—not to say that wellspring of Awesome and Absolutely Annoying (Cloying) Alliteration© and that Mother of all Maddening Mixed Metaphors and Sundry Similitudes®—is back! Yup, like the proverbial phoenix rising from the ashes of near-terminal procrastination, and due to overwhelming popular demand, the W^5/2 is reborn!!! Well, it also might have something...
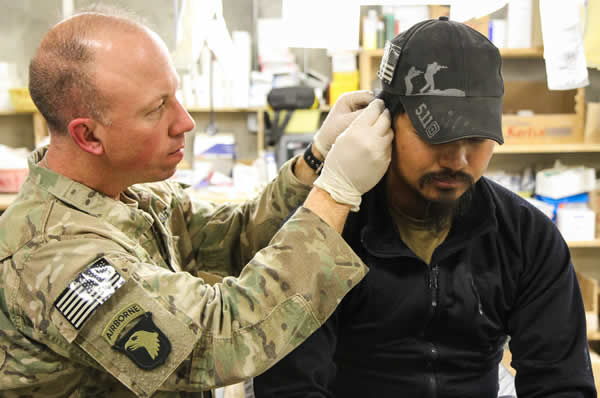
“Battlefield acupuncture”?
THE SCENE: Iraq, Afghanistan, or anywhere where U.S. troops are risking life and limb. THE TIME: The not-too-distant-future. Maybe even 2009. Joe is on patrol. It’s the middle of summer in the desert town. The air hangs heavy, hot, dry and dusty, like a blast furnace firing steel. The heat penetrates Joe’s 80 lb pack in much the same way the heat...

