Category: Pharmaceuticals

Stem cells versus Gordie Howe’s stroke
Note: There is now a major update to this story published here, which explains a lot of the questions remaining in this blog post. Seven years ago I returned to Michigan, where I was born and spent the first quarter century of my life, after an absence of more than 20 years. In the interim, I had done my surgical residency and...

Using the fear of Ebola to promote the placebo legislation that is “right to try”
Libertarians and free market fundamentalists generally detest the FDA and want to dramatically decrease its power in the belief that the free market can guarantee the safety of drugs better than a government agency that requires strong scientific evidence of efficacy and safety before approval. Not surprisingly, they're at it again, this time in the service of promoting "right-to-try" laws and using...
Yahoo News spews NaturalNews anti-vaccine (and other) propaganda
Yahoo News appears to have confused NaturalNews with actual news. It’s not. NaturalNews is the in-house propaganda organ for Mike Adams, whom I’ll introduce in a minute (although he needs no introduction for most readers here). A couple of recent examples: A recycled story, over a year old, from NaturalNews, appearing on Yahoo News last week. It starts out as...
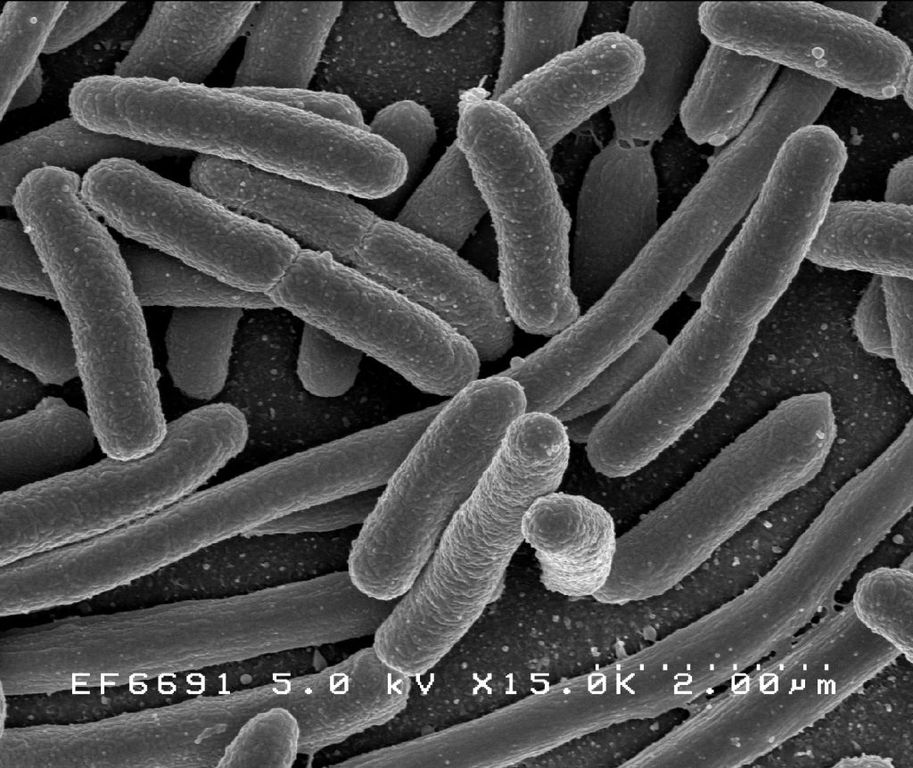
Antibiotics vs. the Microbiome
In 1850, one in four American babies died before their first birthday, and people of all ages died of bacterial infections that could have been successfully treated today with antibiotics. Unfortunately, treatments that have effects usually have side effects, and we are seeing problems due to the overuse of antibiotics. They are given to people with viral infections for which they are...
We’re a drug-taking, supplement-taking nation. So how do we do so safely?
Do you take a vitamin or dietary supplement? It’s increasingly likely that you do, as over half of all American adults took some sort of supplement over the past 30 days. Now there’s evidence to suggest that about one-third of all Americans are taking supplements and prescription drug at the same time, which is renewing questions about risks and benefits. The same...
Lice
If you have children, there’s a good chance you’ve had experience with head lice. Head lice affects as many as 12 million people in the US each year, mostly children. Compared to other health conditions, it is a trivial problem; but it is common and annoying. It can cause itching, notes sent home from school, and often a strong “yuck” reaction. Fortunately,...
Cochrane Reviews: The Food Babe of Medicine?
There are two topics about which I know a fair amount. The first is Infectious Disease. I am expert in ID, Board Certified and certified bored, by the ABIM. The other, although to a lesser extent, is SCAMs. When I read the literature on these topics, I do so with extensive knowledge and, in the case of ID, 30 years of clinical...
Telemedicine: Click and the doctor will see you now
Think you need to see a doctor? How about seeing him (or her) on your computer (or tablet or smart phone) screen instead of in the doctor’s office? The technology of telemedicine, or telehealth, is here. So far, there is no single definition of what it does, and does not, encompass. For example, in some definitions, one of which we discuss today,...
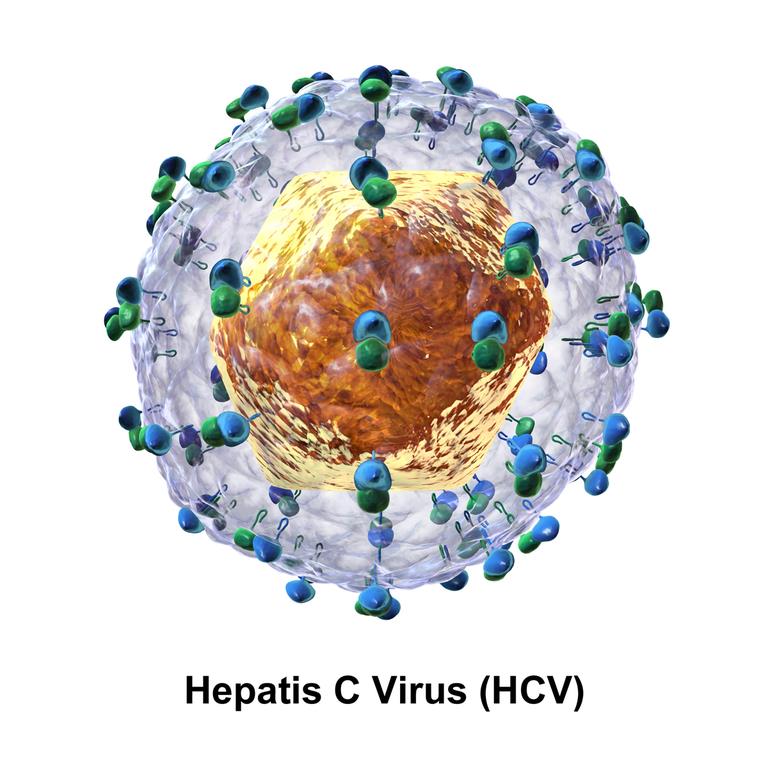
Curing Hepatitis C: A Success Story and a Price Tag
Previously incurable, hepatitis C now has a treatment. A very, very expensive treatment. But there is hope for acutely and chronically infected people.


