Category: Politics and Regulation

HB 4710: Licensing and expanding the scope of practice of acupuncturists in Michigan
Last week, HB 4710, a bill to license acupuncturists, was considered by the Michigan House of Representatives Health Policy Committee. If passed into law, HB 4710 would do far more than license the quackery that is acupuncture. It would also expand the scope of practice of acupuncturists to include homeopathy, "health coaching", and dietary advice, and is yet another example of what...
“Young blood” infusions: same old snake oil
There's no reliable evidence that an infusion of blood plasma from a young donor will benefit an older person, and there are risks, but Ambrosia Health is selling "young blood" infusions for thousands of dollars anyway. The FDA has taken notice.
Dubious for-profit stem cell clinics: Co-opting ClinicalTrials.gov as a marketing tool
Over twenty years ago, cancer quack Stanislaw Burzynski pioneered the abuse of the clinical trial process as a marketing tool to sell his antineoplastons. Now, for-profit stem cell clinics are using ClinicalTrials.gov as a marketing tool for their unproven therapies by listing dubious and scientifically worthless trials in this government database. What can be done?

Walmart sued for deceiving customers in selling homeopathic remedies
A lawsuit claiming Walmart fraudulently deceives consumers in the sale of worthless homeopathic remedies has been filed by the Center for Inquiry (CFI), acting on behalf of the general public. CFI says co-mingling ineffective homeopathic products with science-based treatments on Walmart's pharmacy shelves and website misleads customers into thinking they are equivalent, when "there is not a shred of credible scientific evidence"...
The FDA’s accelerated drug approval program is failing to protect cancer patients
Drug approval is a process that should be and, for the most part, is rooted in rigorous science. However, there is always a countervailing pressure to approve new drugs rapidly, particularly in cancer. That's why the FDA created the accelerated approval program in the early 1990s. Unfortunately, increasingly this approval process appears to be failing us in oncology. Reform is needed.
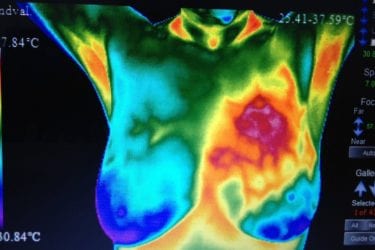
FDA should pull the plug on thermography
The FDA says there's no good evidence that thermography can reliably screen for breast cancer or any other disease. Instead of pursuing the unlawful promotion of thermography for breast cancer detection on a case-by-case basis, the FDA should pull the plug on thermographic devices.

FDA: No CBD in dietary supplements or foods for now, but let’s talk
The FDA reminds everyone that (no matter what your state says) CBD is not a legal ingredient in dietary supplements and foods. The agency is willing to explore changes to the law but unproven claims for CBD health benefits, such cancer cures, will not be tolerated.

Maine considers protecting quacks from accountability to regulators and patients
The Maine Legislature is considering a bill that would put quacks beyond the reach of state healthcare regulatory authorities and leave patients without effective redress for harms.



