Of all the posts I and my cobloggers have written for SBM over the last 15 months, most provoke relatively few comments. However, a few stand out for having provoked hundreds of comments. The very first post that provoked hundreds of comments was Harriet’s excellent discussion of the International Network of Cholesterol Skeptics. In fact, Harriet seems to be quite good at writing posts that provoke a lot of comment, as another of her posts, specifically the one in which she discussed circumcision, also garnered hundreds of comments. However, to my great surprise, the one post that stands out as having received the most comments thus far in the history of SBM is one that I wrote. Specifically, it was a post I called Death by “alternative” medicine: Who’s to blame?, which has collected an astonishing 611 comments thus far. The topic of the post was a case report that I had heard while visiting the tumor board of an affiliate of my former cancer center describing a young woman who had rejected conventional therapy for an eminently treatable breast cancer and then returned two or three years later with a large, nasty tumor that was much more difficult to treat and possibly metastatic to the bone, which would make it no longer even potentially curable. My discussion centered on what the obligation of a physician is to such patients who utterly refuse the science- and evidence-based medicine that we know to be able to cure them of a potentially fatal disease, and I was not only surprised but somewhat taken aback by the vehemence of the discussion.
Since that post, I’ve always been meaning to take a look at what, exactly, the effect of choosing “alternative” medicine over “conventional” medicine is on the odds of survival for breast cancer patients. Even though intuitively one would hypothesize that refusing scientific medicine and relying on placebo medicine instead would have a detrimental effect on survival, it turns out that this question is not as easy to answer as you might think. For example, if you do a search on PubMed using terms like “alternative medicine,” “breast cancer,” and “survival,” the vast majority of the hits will be studies of complementary and alternative medicine (CAM) and breast cancer with little reference to what possible effect these therapies might have on survival. I can envision several reasons for this, the first being that–thankfully–relatively few women actually use alternative medicine exclusively to treat their breast cancer. Also, those that do probably drop off the radar screen of their science-based practitioners, and it is difficult, if not impossible, to capture data regarding their outcomes, given that they all too often stick with their alternative healers until the end. True, they may pop up again in their surgeon’s or primary care doctor’s office with huge, fungating tumors, only to be told that they have to undergo chemotherapy to shrink the tumor before any surgery is possible, after which they will often disappear again. Another important reason is that the natural history of breast cancer is extremely variable, from nasty, aggressive tumors that kill within months to indolent, slow-growing tumors that, even when metastatic, women can survive with for several years. (It is, of course, these women who usually show up in “alternative medicine” testimonials, because they can survive a long time with little or no treatment before their tumors progress.)
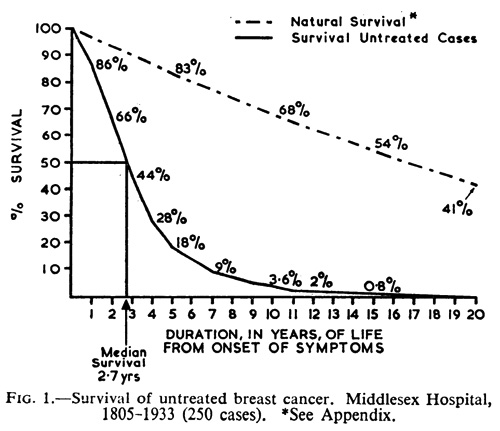
Because it’s important to understand the natural history of breast cancer, I’ll reference a classic study examining the natural history of untreated breast cancer. It was published in 1962 by H. J. G. Bloom, W. W. Richardson, and E. J. Harries, and examined data from Middlesex Hospital from 1805 to 1933 where 250 cases of untreated breast cancer were identified and studied. They calculated survival as the period of time from onset of symptoms to death. What they found was that 18% of the 250 patients survived five years; 3.6% survived 10 years; and 0.8% survived 15 years. Of note, it was 19 years before all patients were dead. Overall, the median survival was 2.7 years. A survival graph from this classic paper is below:
It should be noted that all of these tumors were detected as (at the very minimum) lumps in the breast, given that there was no other way of detecting them at the time. However, the reason we go back to this study time and time again is because, at least in developed countries, it is the rare woman with breast cancer who does not undergo treatment of some kind for it. These days, most tumors are detected at far less advanced stages; indeed, most are detected by mammography. What that means is that, if such a study could be done today, it is very likely that lead time bias would significantly increase the apparent median survival, because increasingly tiny tumors are being found. It is also possible that a significant number of such small tumors may spontaneously regress, which further complicates the issue today, not to mention making it easier to find women who have rejected some or all of “conventional” medicine to treat their cancers and survived significant lengths of time to produce alternative medicine testimonials.
With this background, I have found a couple of studies that can help answer the question. The first one was published in 2005 in the Annals of Surgery by a group in from Geneva University Hospitals. This study involved a search of Switzerland’s database between 1975 and 2000 and included 5,339 patients diagnosed with nonmetastatic breast cancer. The strength of this study is that the Geneva Cancer Registry includes data from all patients from the Geneva canton who underwent treatment and allowed the investigators to compare the outcomes of the women who refused to undergo surgery with curative intent with those who underwent surgery. In the Registry, there were identified 70 patients (1.3%) who refused surgery and concluded:
These women [those who refused surgery] were older, more frequently single, and had larger tumors. Overall, 37 (53%) women had no treatment, 25 (36%) hormone-therapy alone, and 8 (11%) other adjuvant treatments alone or in combination. Five-year specific breast cancer survival of women who refused surgery was lower than that of those who accepted (72%, 95% confidence interval, 60%–84% versus 87%, 95% confidence interval, 86%–88%, respectively). After accounting for other prognostic factors including tumor characteristics and stage, women who refused surgery had a 2.1-fold (95% confidence interval, 1.5–3.1) increased risk to die of breast cancer compared with operated women.
It is true that this is not a randomized study; rather, it is a retrospective study. Consequently, it’s impossible to rule out selection bias, but, as the authors point out, this is one case where doing a randomized study is completely unethical. Moreover, half the women accepted some form of other standard, effective treatm,ent, such as hormonal therapy alone. In any case, what this study shows is that women with no surgery can still live a long time, but are far more likely to die of their cancer than women who do undergo surgical extirpation.
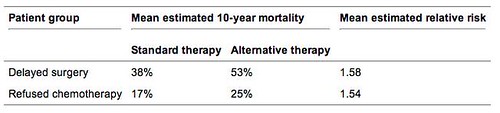
As far as I can find, there is one study that specifically looked at the question of what happens to women who opt for alternative medicine instead of scientific medicine. This study, like the one I just cited, was published in the surgical literature, namely American Journal of Surgery. Given the nature of the question it was seeking to answer, its design is single-armed and retrospective, using prognosis estimated by Adjuvant! Online, an online tool into which clinicians can enter prognostic factors of a breast cancer at the time of presentation and come up with an estimate of chances of survival and recurrence with and without treatment. This, of course, is a weakness, but, again, randomizing patients to scientific medicine or alternative medicine would be completely unethical. In the case of such questions, we scientists have to make do with whatever methodology we can; i.e., do the best we can with what we have. Unfortunately, the study was also small, only 33 patients. Even so, given the huge difficulties involved in undertaking such a study, the investigators, who, as private practitioners operating a community practice in Eugene, OR, went above and beyond the call by trying to look at their data and answer this question. That their study has a number of shortcomings is not their fault; they appear to haved done the best they could with what they had, which includes patients who underwent a panoply of alternative therapies, including coral calcium, herbal therapy, mushrooms, high dose vitamins, whey, chelation therapy, hemlock, and coenzyme Q10.
So what were their findings?
They’re summed up in the following table:
The authors comment:
We found that the overwhelming majority of the patients who initially refused surgical treatment for breast cancer developed disease progression. Five of these patients ultimately underwent surgical resection. Of the other 6 patients, 5 had developed metastatic disease that precluded benefit from surgery. Furthermore, the disease progression caused by the delay in surgery was associated with an increase in the estimated 10-year mortality rate.
Patients who declined chemotherapy or hormone therapy faired slightly better. Optimism for this strategy should be severely tempered by the fact that the length of follow-up evaluation in these patients was relatively short, and these patients had early stage (I or II) disease. By software estimates, the 10-year mortality rate for these patients is still expected to be more than 50% higher than it would have been if the patients had taken their recommended therapy.
A number of patients who expressed their intention to pursue alternative therapies did not return for follow-up evaluation. Attempts were made to contact these patients. Those for whom follow-up evaluation was unavailable were excluded from this study. Although their omission may introduce a selection bias in the results, the effect of this bias is expected to be small because relatively few patients (14 of 47) were in this category.
I find two points important about this study. First, it confirms once again the importance of surgery as a therapeutic modality for breast cancer, especially early stage. Second, and more importantly, it strongly suggests that foregoing or delaying surgery or chemotherapy is at the very least associated with a significantly decreased chance of recurrence-free survival. The authors do note that it is impossible to tell whether this increase in mortality was solely due to delay or refusal of effective therapy or whether the modalities chosen were deleterious. My guess is that it was almost certainly due to the ineffectiveness of the alternative therapies chosen.
More evidence of the uselessness of “alternative” medicine in breast cancer was published two years ago by Edzard Ernst, author of Healing, Hype or Harm? A Critical Analysis of Complementary or Alternative Medicine, which Harriet reviewed about a month ago. In 2006, he wrote a review for the Breast Journal along with Katja Schmidt, MSc, C Psychol, and Michael Baum, MD, ChM, a review entitled Complementary/Alternative Therapies for the Treatment of Breast Cancer. A Systematic Review of Randomized Clinical Trials and a Critique of Current Terminology. The objective of the study was to examine all studies randomized clinical trials (RCTs) for “alternative cancer cures” (ACCs). Treatments examined included various methods of psychosocial support such as group support therapy, cognitive behavioral therapy cognitive existential group therapy, a combination of muscle relaxation training and guided imagery, the Chinese herbal remedy Shi Quan Da Bu Tang, thymus extract, transfer factor, melatonin, and factor AF2.
The first finding was that the methodological quality of the studies was, by and large, pretty low. The most common deficiencies included: lack of power sample calculation; small sample size; lack of adequate randomization and/or (patient and assessor or only assessor) blinding; and insufficient follow-up periods. It was noted that only one trial applied an intention to treat analysis. From the 15 studies Ernst examined, this is what was concluded:
The totality of the data fails to show a single intervention that would be demonstrably effective as an ACC. The paucity and the often-low methodological quality of the RCTs are as unexpected to us as they are disappointing. Most trails had small sample sizes; thus a type II error is conceivable. But even if this were true, one would be correct in stating that to date, no effective ACC has been identified.
A lot of this is, of course, true based on discussions of prior plausibility alone. One could argue that, given the poor quality of the studies examined by Ernst, there might be an effect that was missed. However, if an effect were missed, it would have to be small or, at most, moderate. That is not what is claimed for many of these ACCs. What is often claimed is a near-miraculous “cure” for cancer, which, if it were true, would be relatively easy to detect. As I’ve often argued about, for example, the Gonzalez regimen for pancreatic cancer, if such ACCs really were cures, it would actually be fairly easy to show. In the case of pancreatic cancer, for instance, just producing well documented case reports of a few five year survivors among patients with documented metastatic adenocarcinoma of the pancreas would, I daresay, make even Wally Sampson and Kimball Atwood sit up and take notice. Somehow, we never see this. Of course, what makes the question in breast cancer more difficult to answer is its highly variable natural history. In contrast, the vast majority of patients with pancreatic cancer die within the first year (more than half die within six months); fewer still live beyond two years; and very close to none live beyond three years. In contrast, lots of women with metastatic breast cancer live longer than two or three years; a few.
Finally, about five years ago, there was a study out of Norway that looked at the effect of alternative medicine on cancer survival. This study did not limit itself to breast cancer, but it is interesting and useful nonetheless. The hypothesis of the study was that the use of alternative medicine does not have any effect on the survival of cancer patients, and to test the hypothesis investigators studied data from surveys done by the Norwegian Board of Health, which asked patients standard demographic information, but then asked them about their use of alternative medicine as follows:
In the questionnaire presented to the patients, AM was defined as any treatment outside of mainstream therapy that had been used to treat their cancer. A multiple-choice list consisting of the best known and frequently used non-proven methods in Norway was presented. Patients also had the opportunity to add other types of alternative therapy in response to an open question.
The following alternative methods were described in the multiple choice questionnaire: Use of biological treatments, herbs, faith healing or healing by hand, homeopathy, reflexology (zone therapy), megadoses of vitamins, diet treatments, injection therapies such as iscador (a mistletoe preparation) and a Norwegian injection therapy called “Nitter therapy”. Nitter therapy consists of vitamin B12, gammaglobulins, tranexamic acid, multivitamins and nutritional supplements.
Modalities such as relaxation, psychotherapy, participation in a self-help group or changes in lifestyle activities that were used to reduce distress and to improve the patient’s subjective well being were not coded as AM.
I actually very much approve of the fact that the investigators did not define relaxation, psychotherapy, lifestyle changes, etc., as “alternative.”
A total of 515 patients were analyzed, and results were as follows:
In January 2001, survival data were obtained with a follow-up of 8 years for 515 cancer patients. A total of 112 (22%) assessable patients used AM. During the follow-up period, 350 patients died. Death rates were higher in AM users (79%) than in those who did not use AM (65%). In a Cox regression model adjusted for demographic, disease and treatment factors, the hazard ratio of death for any use of AM compared with no use was 1.30, (95% Confidence Interval (CI) 0.99, 1.70; P=0.056), suggesting that AM use may predict a shorter survival. Sensitivity analyses strengthened the negative association between AM use and survival. AM use had the most detrimental effect in patients with an ECOG (Eastern Cooperative Oncology Group) performance status (PS) of 0 (hazard ratio for USE=2.32, 95% CI, 1.44, 3.74, P=0.001), when compared with an ECOG PS of 1 or higher. The use of AM seems to predict a shorter survival from cancer. The effect appears predominantly in patients with a good PS.
In other words, not surprisingly, use of alternative medicine is correlated with poorer surival in the patients with a good performance status and thus who are likely to have a more favorable prognosis. Patients with a poor performance status are more likely to undergo less aggressive therapy because they are less able to tolerate, for example, radical surgery or heavy-duty chemotherapy regimens.
The authors speculated about a number of reasons why use of alternative medicine might be associated with poorer survival. Obviously, it may not be causative. (Remember again that correlation does not necessarily equal causation. One possible explanation is that users of alternative medicine are less likely to undergo optimal medical therapy. One interesting possibility is that users of alternative medicine were more likely to be having severe symptoms, which led them to turn to “alternative medicine” to try to relieve their symptoms. In any case, this study had a number of problems, including a high attrition rate that could have resulted in significant selection bias.
Putting it all together, I conclude that there is no compelling evidence for a significant survival benefit due to any “alternative” therapy, nor is there good evidence for significant treatment effects. The studies that do purport to show an effect are virtually all plagued with methodological difficulties and tend to show effects that are barely above background noise. Even the much touted psychosocial support of late has failed to demonstrate any improvement in the survival of cancer patients. Moreover, although there is a relative paucity of studies, and they, too, are generally retrospective and difficult to interpret, what evidence is out there is that alternative medicine use among cancer patients is associated with an increased risk of dying from cancer, particularly when conventional therapy is eschewed. Taken together, these data make it very hard not to conclude that at best the vast majority of alternative therapies are either useless, no more than placebos, or might even be harmful. That is why they have no role in science-based medicine at present.
The idea of an “alternative cancer cure” assumes that conventional oncology would not adopt a cancer treatment simply because it originates from an area outside of mainstream medicine. We feel that, should such a cure one day emerge, it would be investigated without delay by oncologists and adopted into routine care as soon as the data supporting it are convincing. Plant-based cancer medications such as Vincristin and Vinblastin (both extracted from the plant Vinca rosea) or Taxol (Taxus baccata) could be employed to back up this theory. It follows that the term ACC is and most likely will always be a contradiction in terms.
Or, as I frequently put it: There is no such thing as “alternative” medicine. There is medicine that is effective, medicine that is not, and medicine that has not been tested yet. Nearly all of so-called “alternative” medicine falls into one of the latter two categories, and those that have not been tested yet nearly all fall into the category of being so wildly improbable that testing them without more positive evidence makes no sense. In any case, as a cancer surgeon, I don’t care where a therapy came from. I really don’t. If someone could show me that reiki or homeopathy cures cancer, I’d use either. In the meantime, I will continue to argue that the very concept of “alternative” medicine is a potentially deadly false dichotomy for cancer patients.
REFERENCES:
- H. J. G. Bloom,, W. W. Richardson, & E. J. Harries (1962). Natural History of Untreated Breast Cancer (1805-1933) British Medical Journal, 2, 213-221 DOI: PMC1925646
- Chang, E., Glissmeyer, M., Tonnes, S., Hudson, T., & Johnson, N. (2006). Outcomes of breast cancer in patients who use alternative therapies as primary treatment The American Journal of Surgery, 192 (4), 471-473 DOI: 10.1016/j.amjsurg.2006.05.013
- T Risberg, A Vickers, R.M Bremnes, E.A Wist, S Kaasa, & B.R Cassileth (2003). Does use of alternative medicine predict survival from cancer? European Journal of Cancer, 39 (3), 372-377
- Verkooijen, H., Fioretta, G., Rapiti, E., Bonnefoi, H., Vlastos, G., Kurtz, J., Schaefer, P., Sappino, A., Schubert, H., & Bouchardy, C. (2005). Patients’ Refusal of Surgery Strongly Impairs Breast Cancer Survival Annals of Surgery, 242 (2), 276-280 DOI: 10.1097/01.sla.0000171305.31703.84