
The closure of major integrative medicine “Crown Jewels”: Terminating the Terminator?
When it comes to expansion and infiltrating medicine, "integrative medicine" has frequently seemed like the Terminator: utterly relentless. Recent setbacks at major integrative medicine "Crown Jewels" resulting in their closure cast that narrative in doubt. However, I never forget that after its seeming destruction, the Terminator always comes back.

Quackery for Kids
A brief rant and a few random observations on quackery for kids.
A closer look at generic drugs
In the struggle to contain growing healthcare costs, generic drugs have the potential to save millions. What gives us confidence they are equivalent to brand-name drugs?
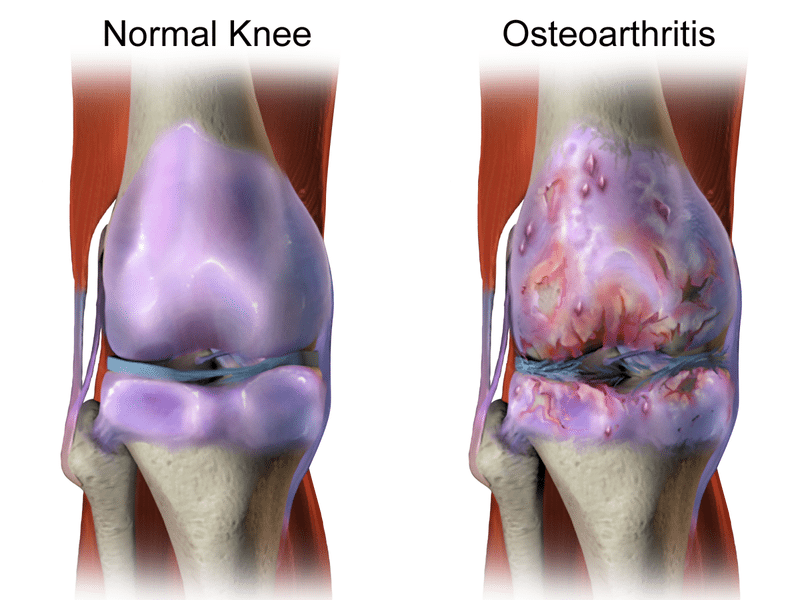
New Evidence for Chondroitin
Several previous studies showed chondroitin was ineffective for knee osteoarthritis, but a new study says it is as effective as celecoxib. There are reasons to be skeptical.

Medical marijuana as the new herbalism, part 5: Turning herbalism into science-based medicine
There’s a new clinical trial published in the New England Journal of Medicine showing a beneficial effect due to cannabidiol, a chemical isolated from marijuana, on drug-resistant seizures due to Dravet syndrome. Although there are a fair number of caveats, this is how you begin to turn the herbalism that characterizes medical marijuana advocacy into science-based medicine.

Corrigendum. The Week in Review for 05/28/2017.
More measles in Minnesota. Yet another form of acupuncture: Snafu. CAM cancer studies are of poor quality. The future of CAM is LSD.

I Got Nothing
Really nothing here from me this week. Go have a beer. Time better spent.

Healthcare reform should ditch mandated coverage of CAM providers
Forced insurance coverage of chiropractic, naturopathic, and acupuncture services is not consistent with the goals of either the ACA or the AHCA. Whatever happens to Obamacare in the U.S. Senate, Section 2706 of the ACA should be repealed.

The Natural Cancer Cure Narrative
Julie Reason and her husband are producing a documentary about her cancer, and efforts to cure it. Based on their comments, they are drawing upon an established and false narrative about the causes and cures of cancer, one that can be dangerous to her, and all other cancer patients.





Mast Cell Activation Disorder – Yes, It’s Real
Mast Cell Activation Disorder is real, but there are a large number of fake diagnoses out there. How do you tell the difference?