
Bee Venom is Snake Oil
Bee venom acupuncture is a double-barrel pseudoscience that provides new example of an old problem - the use of poor quality preclinical research to justify the inclusion of nonsense in medicine.
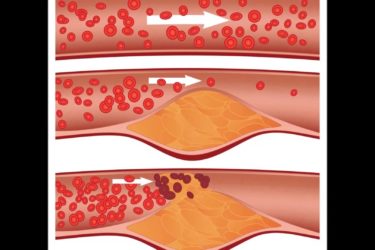
New Study Finds that Statins Prevent Cardiovascular Deaths
A new meta-analysis shows that statin drugs are effective at preventing cardiovascular events and death, especially in patients with a baseline LDL cholesterol >100.
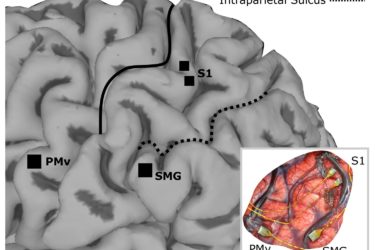
Adding Sensation to Robotic Limbs
Recently scientists have managed to stimulate the brain in such a way that approximated some of the sensations of a natural limb in a paralyzed subject. No, they did not regain sensation, but the research is a powerful proof of concept. It shows that it is possible to produce natural-feeling sensation through electrical stimulation of the cortex, an important step for brain-machine...
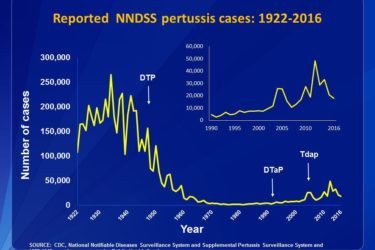
The Pertussis Resurgence
A new analysis shows that the resurgence of pertussis is largely due historical patterns of vaccination. This and other data show the importance of full vaccine compliance in preventing returning epidemics of this deadly disease.
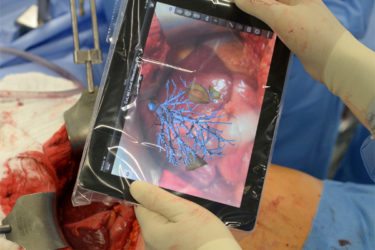
Augmented Reality in Medicine
Augmented reality has the potential to revolutionize how physicians access data while caring for patients, whether in the operating room or clinic.
Music for ADHD?
In a recent "Ask Me Anything" on Reddit, the CEO of Brain.fm claimed his company's music can improve concentration and help with ADHD. At the very least I see such claims as highly implausible, and not something we can conclude from the existing basic science research. I have no problem with doing clinical research, and maybe we might learn something about how...

Facilitated Communication Rears Its Ugly Head Again
Facilitated communication is pure pseudoscience that was debunked almost 30 years ago, but it keeps coming back, creating new victims.
Too Many Too Soon? No!
The results of a study looking at, in part, the "too many, too soon" complaints of antivaccination activists were completely negative. There was no difference in vaccine antigen exposure between two groups who differed in the number of infections over a two year period. Therefore there is no correlation between vaccine antigen exposure and susceptibility to other infections.
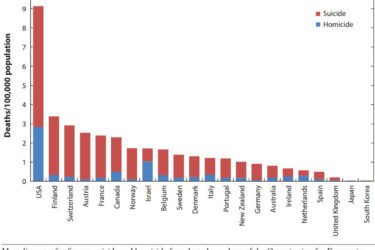
Gun Violence as a Public Health Issue
Gun violence is a serious public health issue in America but is not getting the research that it deserves. This needs to change.


Where Are We With the Replication “Crisis”
The replication problem is not as bad as the sensational reporting has suggested. But it is still a legitimate issue that needs to be addressed.