One of the recurring topics here at SBM is the idea of the placebo: What it is, what it isn’t, and how it complicates our evaluation of the scientific evidence. One my earliest lessons after I started following this blog (I was a reader long before I was a writer) was that I didn’t understand placebos well enough to even describe them correctly. Importantly, there is no single “placebo effect”. They are “placebo effects”, a range of variables that can include natural variation in the condition being studied, psychological factors and subjective effects reported by patients, as well as observer bias by researchers studying a condition. All of these, when evaluated in clinical trials, produce non-specific background noise that needs to be removed from the analysis. Consequently, we compare between the active treatment and the placebo to determine if there are an incremental benefits, to which we apply statistical tests to determine the likelihood that the differences between the intervention and the placebo groups are real. Removed from the observational nature of the clinical trial, we can’t expect the observed “placebo effects” to persist, as they’re partially a consequence of the trial itself. A more detailed review of placebos is a post in and of itself, so I’ll refer you to resources that describe why placebo effects are plural, that placebo effects are subjective rather than objective and there is no persuasive evidence to suggest that placebo effects offer any health benefits. What’s most important is the understanding that placebo effects are a measurement artifact, not a therapeutic effect.
Placebo effects are regular topics within in SBM posts because an understanding of placebo effects is essential to evaluating the evidence supporting (so-called) complementary and alternative medicine, or CAM. As better quality research increasingly confirms that the effects from CAM are largely, if not completely, attributable to placebo effects, we’ve seen the promoters of CAM shifting tactics. No longer able to honestly claim that CAM has therapeutic effects, “treatments” such as acupuncture or homeopathy are increasingly promoted as strategies that”harness the power of placebo” without all the pesky costs or side effects of real medical interventions. But this is simply special pleading from purveyors and promoters. Unable to wish away the well-conducted trials that show them to be indistinguishable from placebos, they instead are spinning placebo effects as meaningful and worthy of pursuit – ideally with your favourite CAM therapy. Again, I’ll refer you to posts by David Gorski and Steven Novella who offer a more detailed description of how negative results can be spun to look positive. Because CAM’s effects are indistinguishable from placebo, we should not invest time and resources into pursuing them – we should instead focus on finding treatments that are demonstrably superior to placebo.
But what if physicians are already using placebos widely in practice? Setting aside the ethical issues for now, widespread placebo usage might suggest that physicians believe that placebos are effective treatments. And that’s the impression you may have had if you skimmed the medical headlines last week:
97 percent of UK doctors have given placebos to patients at least once said the press release
Doctors like to ‘sugar the pill’ said The Express
Most doctors prescribe placebos – said The Telegraph
Probing the power of placebos – The Globe and Mail
All of this comes from survey published published in PLOS ONE last week, Placebo Use in the United Kingdom: Results from a National Survey of Primary Care Practitioners. Before diving into the findings, let’s consider the context of placebo use – not in clinical trials, but in the real world.
Is it a placebo in the eye of the prescriber?
The deliberate use of inert therapies in medicine is not a new topic and has been covered before at SBM. Like other contributors to this blog, I find the idea of actively prescribing placebos to be both unethical and highly paternalistic. Why jeopardize patient trust? Back in 2011 I summarized the evidence on the prevalence of placebo prescribing. But to understand the data we need to define “placebo” when it’s being used outside a clinical trial. Not every placebo is a sugar pill, and not every placebo is used with “placebo” intent. There are two forms of placebo in routine medical use: “Pure” placebos are truly inert: they contain no medicinal ingredients. These are the sugar pills, saline injections, and most homeopathic “remedies”. They’re the products we used in clinical trials when it’s “placebo-controlled.” “Impure” placebos, on the other hand, actually contain medicinal ingredients, but are ineffective for the condition being treated. This could be because of a sub-therapeutic dose, or the active ingredient has no effectiveness against the condition being treated (e.g., antibiotics for viral infections). We don’t use impure placebos in clinical trials, as there’s the risk it could cause its own effects or adverse events. Pure or impure, a placebo must be ineffective for the condition being treated – otherwise it’s not a placebo.
There is a lack of good data that summarizes on how routinely placebos are deliberately prescribed. A 2010 paper provides the most comprehensive summary, and the findings are complicated by inconsistent questions, changing definitions of what a placebo actually is, and other study design problems. Consequently the results of reported placebo use by physicians varies: 17-80% for pure placebos, and 41-99% for impure placebos, in response to “have you ever prescribed” questions. But few physicians use placebos regularly, and “impure” placebos are used far more frequently used than “pure” placebos. Which is understandable, as prescribing antibiotics for what’s likely (but not unequivocally) a viral infection should be more common than the deliberate use of sugar pills and deception to make a patient think they’re getting medicine when they’re not.
So let’s take a closer look at the most recent study, the one that generated all of the headlines. As it’s at PLOS ONE, the full text is available. The lead author was Jeremy Howick, a PhD scientist, and the study was funded by The University of Oxford Department of Primary Care Health Sciences and The Southampton Complementary Medical Research Trust. “Complementary”? Howick writes under the name “Dr. Placebo” at trusttheevidence.net, and his writing suggests that he believes placebos have physiologic effects. In fact, Howick goes so far as to suggest that given the side effects of real drugs, “not prescribing placebos might be even less ethical.” Another of the coauthors is George Lewith, a “complementary medicine” physician who has published research on homeopathy and is a proponent of “integrative” healthcare. Lewith has an equally interesting perspective on the use of placebos in clinical practice, one that we’ll come back to, when we interpret the findings of the survey and the conclusions drawn by the authors.
This was a web-based survey of over 1700 general practitioners registered with a marketing service. 783 completed the questionnaire, which is an fair response rate for a survey, but could bias the sample towards those interested in placebos. The sample that responded was 55% male, in practice for an average of 20 years – so these were not new graduates. The way the authors describe the rationale for the study belies their interest “complementary” medicine:
Since this was an empirical rather than conceptual study we adopted a pragmatic approach and asked doctors whether they used various treatments described as placebos in other similar surveys. Our approach has the advantage of being useful: patients, doctors, and policy makers care more about whether particular treatments are effective and ethical than whether these treatments carry the label ‘placebo’.
Rather than useful, the “pragmatic” approach they used gives results that are the opposite of useful – they’re highly biased. As I noted above, the systematic review by Fässler illustrates that different definitions of “placebo” can have a significant influence on the results reported. To their credit, the authors use a “pure placebo” definition that’s consistent with other studies: “sugar pills or saline injections without direct pharmacologically active ingredients for the condition being treated.” But their definition of “impure placebo” is so expansive that it’s not clear to me that the authors understood what an “impure placebo” is supposed to be:
- Positive suggestions
- Nutritional supplements for conditions unlikely to benefit from this therapy (such as vitamin C for cancer)
- Probiotics for diarrhea
- Peppermint pills for pharyngitis
- Antibiotics for suspected viral infections
- Sub-clinical doses of otherwise effective therapies
- Off-label uses of potentially effective therapies
- Complementary and Alternative medicine (CAM) whose effectiveness is not evidence-based
- Conventional medicine whose effectiveness is not evidence-based
- Diagnostic practices based on the patient’s request or to calm the patient such as
- Non-essential physical examinations
- Non-essential technical examinations of the patient (blood tests, X-rays)
There are some reasonable inclusions. Subclinical doses are also appropriately categorized as “impure” placebos, as are products that are ineffective for the condition being treated (e.g., antibiotics for suspected viral infections). But there are some odd entries, too.
Positive suggestions are considered an “impure placebo” yet they can be part of routine patient encounters and not offered with deliberate placebo intent. I give positive suggestions to patients routinely when I reassure them their cold is self-limiting and will resolve in few days, medicated or not. This isn’t an “impure placebo”. Probiotics for diarrhea lack a strong evidence base but may not be given with placebo intent, as practitioners may believe they are effective. Bizarrely, all off-label drug use is considered “placebo”, suggesting either the authors have very little practical knowledge of medical practice, or are deliberately trying to widen the category of “impure placebo”. Off-label use means a drug is unapproved for the intended use. It does not indicate there is a lack of evidence, only that a regulator hasn’t formally approved use. When drugs are used for niche purposes (e.g., pediatric prescribing or rare diseases) pharmaceutical manufacturers may not bother seeking regulator approval. Bizarrely, they also include “conventional medicine whose effectiveness is not evidence based” as a separate category. Finally, the inclusion of CAM “whose evidence is not evidence-based” is telling. CAM which is evidence-based isn’t CAM any more – it’s simply medicine. And ineffective CAM may not be a placebo, as ineffective doesn’t mean safe, or free of side effects. Diagnostic practices are also included here as “impure placebos” which stretches the definition of what a a placebo is. Physical exams are not placebos – they may be used to rule out a diagnosis or to reassure a patient about the certainty associated with a diagnosis. They’re not expected to have therapeutic effects, but may be done to comfort patients, or reassure them of the diagnosis. Similarly, lab tests are not “placebo” treatments – no-one expects a therapeutic benefit from an X-ray, but may be reassured by the results. They may be completely unnecessary, but they’re not a therapeutic placebo. Again – a placebo must be ineffective for the condition being treated – otherwise it’s not a placebo.
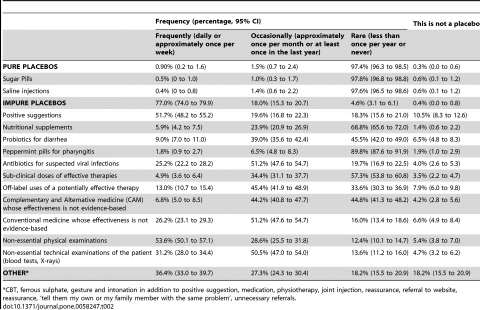
To their credit, the investigators did offer respondents the opportunity to indicate that an intervention was “not a placebo”. But on the primary question of placebo use, we should expect the use of impure placebos to be off the charts. And that’s exactly what we see in the results. Here’s the results to the primary question:
12 % reported using pure placebos at least once in their career and 97% reported using impure placebos at least once. Given the definitions, it should not be surprising that nearly every physician has used what the authors defined as an “impure” placebo at least once.
When we break down the placebo use, the results are a bit more useful:
The frequency at which pure placebos are used frequently or occasionally is very low – consistent with other studies. 97.4% use pure placebo less than once per year, or have never used a pure placebo.
The breakdown of “impure” placebo is much more interesting than what the headlines reported. Not surprisingly, positive suggestions were the most commonly reported (71% reporting frequent or occasional use) with 11% indicating it wasn’t a placebo at all. Beyond that, few were used frequently with the exception of antibiotics for suspected viral infections (75% saying frequently or occasionally) and medicine used in a non-evidence-based way (77%). Non-essential physical exams were the most common non-therapeutic offered (54% reporting frequently), but again, I don’t feel this is a placebo treatment – but only 5% of physicians apparently agreed with me.
Then there’s the “other” category which again left me wondering what isn’t considered a placebo in this study, as cognitive behavioural therapy, iron tablets, physiotherapy, reassurance and even “referral to website” are included as well.
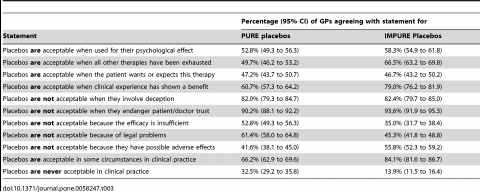
Finally physicians were asked a series of questions about situations where placebo use would be considered acceptable and unacceptable:
Physicians were largely split on their acceptability of placebo for psychological effects, or when “the patient wants or expects this therapy”. In response to their acceptability “when clinical experience has shown a benefit” seems carefully worded to dodge the reality of placebo and instead appeal to anecdotes – “clinical experience”. In this case, the reported acceptability climbs to 60% for pure placebos and 79% for impure placebos. Reassuringly, there were strong opinions that placebos are not acceptable when they involve deception or may jeopardize patient trust. Perhaps not surprisingly the authors call these ethical constraints “perhaps sometimes unjustified” in the discussion section of the paper.
A nation of placebo prescriptions?
The authors go for the headlines and paint a picture of widespread placebo use in their discussion of the results:
Placebos may represent one of the more commonly used treatments in UK primary care in spite of (perhaps sometimes unjustified) ethical constraints. Twelve percent of respondents reported using pure placebos, and 97% reported using impure placebos at least once in their career. Many placebos were used frequently by over half the respondents, and most general practitioners felt there were circumstances in which impure and pure placebos were ethically acceptable. Half of the practitioners who use placebos informed their patients that this intervention has helped other patients without specifically telling them that they were prescribing a placebo. This raises unresolved ethical issues about how GPs approach informed consent in relation to their prescriptions of placebos.
My interpretation is a bit different. The survey was explicitly designed to make placebo use appear widespread. A closer look suggests that physician behavior is more nuanced and circumspect than what the headlines suggested. Regular prescribing with pure placebos is exceptioanlly rare. Physicians agree that they are not acceptable when they involve deception or endanger patient trust. In some situations, they feel they’re acceptable. But this clearly seems to be the exception, rather than the rule. On balance, it’s what I’d expect in a physicians or health professional.
However, given the background of the authors, and their interest in promoting the active use of placebo treatments, the paper has a different conclusion:
The time has come to use similar methods to investigate ways to rationalize placebo use. The long term viability of placebo use in clinical practice depends on whether placebo benefits outweigh harms, their cost, and whether patients and practitioners deem their use to be ethically acceptable. Further investigations are warranted to develop ethical and cost-effective placebos.
I’m sure the authors have some ideas about just what those ethical and cost-effective placebos might be. The design and results seem perfectly designed to position the use of CAM to generate what they believe are clinically relevant placebo effects. In media quotes, the intent is obvious:
“This is not about doctors deceiving patients,” said Dr Jeremy Howick, co-lead author of the study from the University of Oxford. The study shows that placebo use is widespread in the UK, and doctors clearly believe that placebos can help patients.”
Professor George Lewith, co-lead author of the study from the University of Southampton, said: “Other previous published studies by Southampton have clearly shown placebos can help many people and can be effective for a long time after administration. The placebo effect works by releasing our body’s own natural painkillers into our nervous system. In my opinion, the stigma attached to placebo use is irrational, and further investigation is needed to develop ethical, cost-effective placebos.”
The SKEPT!CAL blog ran the press coverage through the churnalism.com website and, disappointingly, found the media largely reported the press release nearly verbatim.
Conclusion
Given what appears to be an established interest by at least some of the authors in promoting the routine use of placebos and placebo treatments in medical practice, the design and results of this study should surprise no one. The survey was designed to give the impression of widespread, active placebo use by UK physicians. That’s what they found, that’s what they announced, and that’s what the media reported. The authors used this carefully designed argument from popularity to support their statements about the therapeutic legitimacy of placebo usage. Finally, add in some spin to suggest that placebo effects are therapeutically meaningful. And to what end? Because if physicians are already using placebos routinely, why not fund a few more – like…say…CAM? I’m sure the Southampton Complementary Medical Research Trust can think of a few products that might fit the bill.
The reality, of course, is quite different, and the survey results reflect this. Physicians use placebos relatively infrequently and generally recognize that they are unacceptable when they involve deception and jeopardize patient trust. Quite frankly, it’s not worth that risk. The most we can hope for when we use placebos are subjective reports of changes in a patient’s symptoms. Placebos have no objective, clinically meaningful effects. When a drug cannot be show to be more effective than placebo, we stop using the drug – we don’t repackage the placebo as a therapeutic agent. But that’s exactly what placebo proponents are proposing.
Reference
Howick J., Bishop F.L., Heneghan C., Wolstenholme J., Stevens S., Hobbs F.D.R., Lewith G. & Manchikanti L. (2013). Placebo Use in the United Kingdom: Results from a National Survey of Primary Care Practitioners, PLoS ONE, 8 (3) e58247. DOI: 10.1371/journal.pone.0058247.t003