Category: Herbs & Supplements

Amish Farmer Jailed for Selling Snakeoil
An Amish farmer is convicted of selling a caustic poison as patent medicine (and of witness tampering) and yet is defended by "alternative medicine" proponents who apparently want the freedom to be defrauded and harmed.

Adrenal fatigue: A fake disease (updated)
“Adrenal fatigue” is not a real medical condition, but some claim it is real and that medicine is ignoring it. What does the science say?

Coconut Oil Warning
Coconut oil is promoted as a health food, but a recent warning from the American Heart Association warns that coconut oil is very high in saturated fats and increases your risk for heart disease.

On Guard, DōTERRA, Essential Oils, and a Lesson in Reading Research Studies
A study of On Guard™, a mixture of essential oils, showed that it reduced the infectivity of influenza virus in dog kidney cells in the lab; but that's irrelevant to the question of whether the product has any clinical effect in humans.
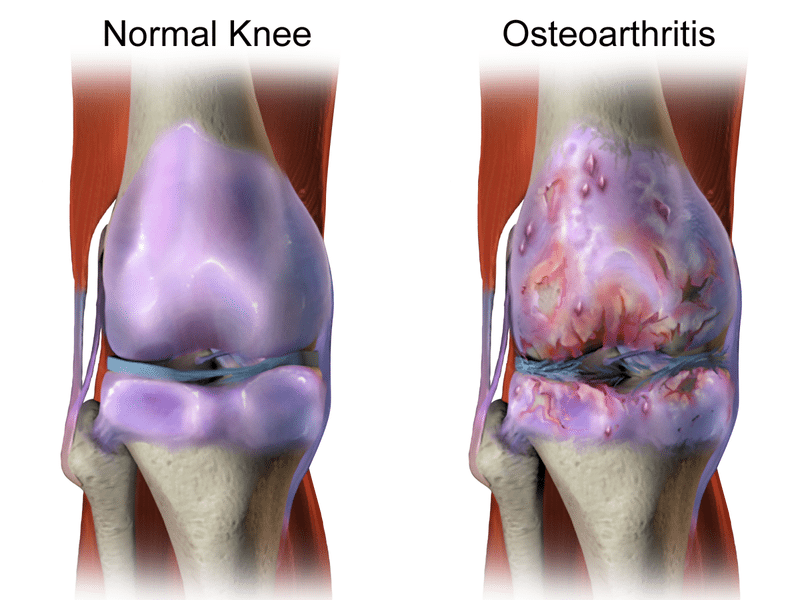
New Evidence for Chondroitin
Several previous studies showed chondroitin was ineffective for knee osteoarthritis, but a new study says it is as effective as celecoxib. There are reasons to be skeptical.

Medical marijuana as the new herbalism, part 5: Turning herbalism into science-based medicine
There’s a new clinical trial published in the New England Journal of Medicine showing a beneficial effect due to cannabidiol, a chemical isolated from marijuana, on drug-resistant seizures due to Dravet syndrome. Although there are a fair number of caveats, this is how you begin to turn the herbalism that characterizes medical marijuana advocacy into science-based medicine.

Protandim Update: New Studies and an FDA Warning Letter
Multilevel distributors of the dietary supplement Protandim think that evidence from scientific studies supports their claims for their product. The FDA disagrees.

The Medical Medium’s Thyroid Pseudoscience
Anthony William calls himself a "Medical Medium". He has no medical expertise, but he provides medical advice based on claimed communication with the spirit world. What could possibly go wrong?

Medical Neglect of Children
Medical neglect caused horrific suffering for these children, ending in death or permanent impairment. Their parents failed them, but so did society.


