Category: Legal
Corrigendum. The Week in Review for 04/09/2017
The NECSS is coming. Acupuncturists mimic chiropractic. Flu vaccine prevents death. In the UK they care more for cats than people. The problem is my middle burner, not too many burgers. And more.
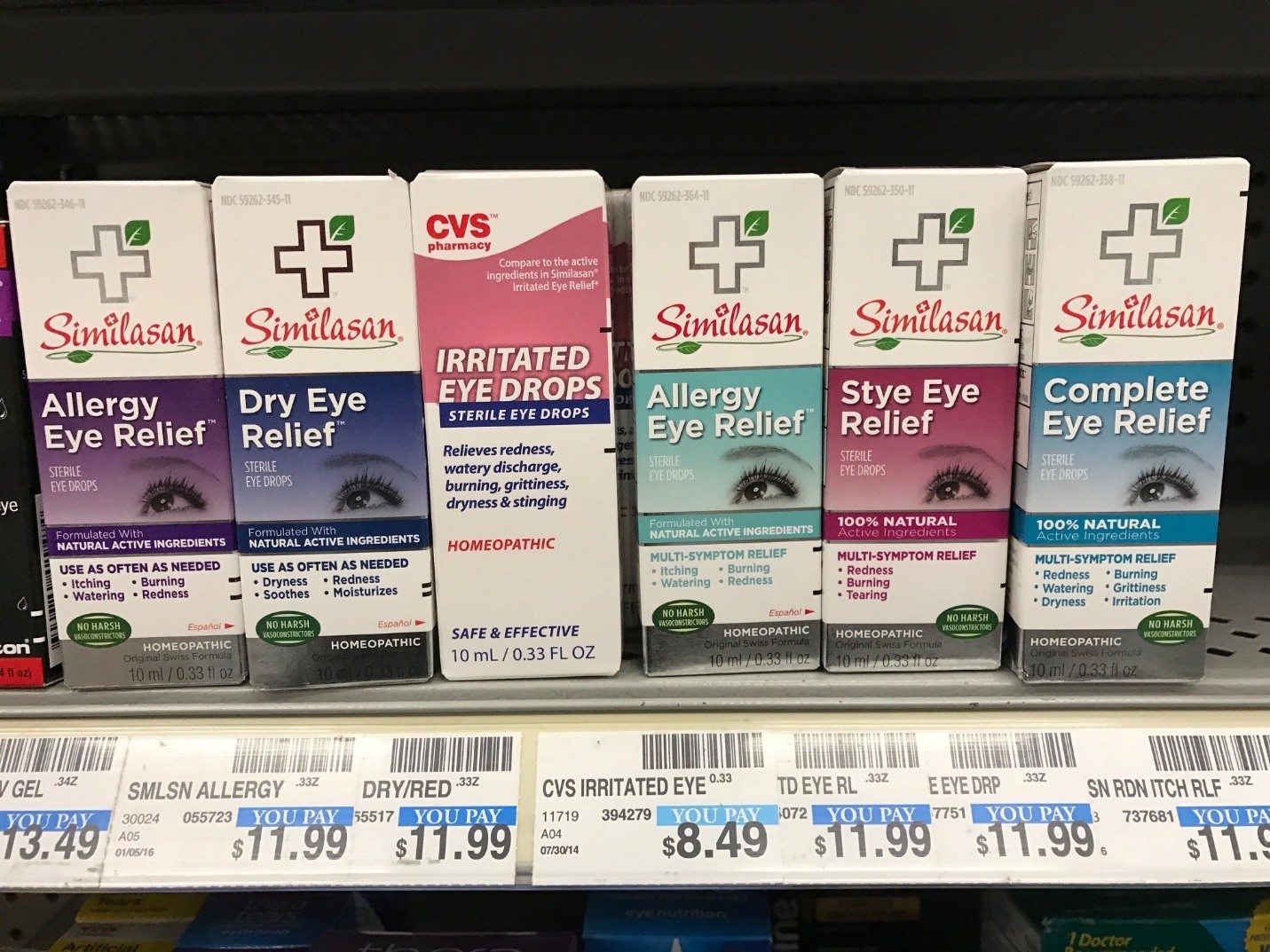
CVS selling homeopathic remedies: It gets personal
I almost purchased a worthless homeopathic eye remedy at CVS for a cancer patient. I'm taking action to try to stop this from happening to others.

Health Savings Accounts and Quackery Revisited
Health savings accounts don't require medical treatments to be safe or effective for consumers. This leads to taxpayer-supported quackery.

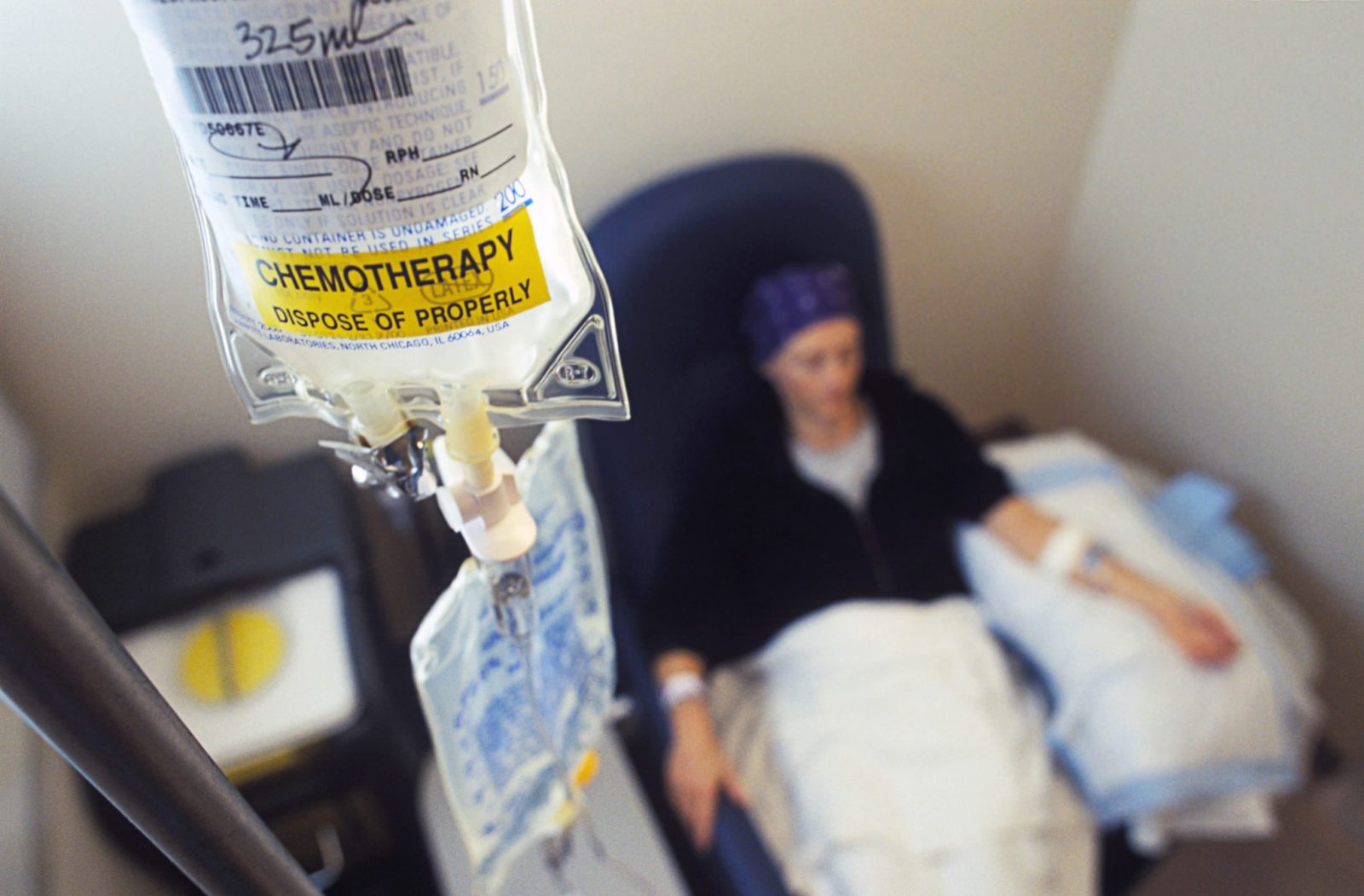
“Chemotherapy is for losers”: A tragic tale of cancer, naturopathic quackery, and murder
When a patient and her family trust a naturopath rather than oncologists and oncologic surgeons, the result is often tragic. In this case, Fikreta Ibrisevic trusted naturopath Juan Sanchez Gonzalez instead of real doctors to treat her rhabdomyosarcoma in 2015. The results were as tragic as expected, and she died. What happened next was not expected and amplified the horror of the...
Bills remove impediments to ill-advised state “right to try” laws, shield wrongdoers, and hide adverse events
Congressional bills will unleash state "right to try" laws, block terminally ill patients from redress for damages caused by negligent doctors and drug companies, and hide adverse drug events from the public.

Corrigendum. The Week in Review for 02/19/2017
More poorly done acupuncture studies. Burzynski eats just desserts. Italians like homeopathy. New Jersey is going after Oregon. And more
Turning chiropractors into primary care physicians via Legislative Alchemy
Via the magic of Legislative Alchemy, chiropractic lobbyists are trying to to convince state legislators to expand chiropractic scope of practice so they can rebrand as primary care physicians.
Corrigendum: The week in review for 2/5/2017
Drinking hydrogen peroxide kills. Homeopaths don't care if their nostrums kill children. Acupuncture is placebo. But you knew that. Saudi Arabia bans reiki for the damnedest reasons. Eating placentas. And more!

Board Disciplinary Actions. What Naturopaths Really Do Not Want You To Know
Disciplinary actions against ND's in Oregon by the Board. How to find them and what they are.

Corrigendum. The week in SBM for 1.22.2017
Not every article and study that pops up my feeds in the world of pseudo-medicine is worthy of a complete blog post. But they need to be noticed and commented upon: Liver toxicity from herbs. Popped lungs from acupuncture. Chiropractic does not help scoliosis. Yoga is just exercise. There are eight kinds of wind: Great Feathery Wind, Scheming Wind, Hard Wind, Great...

