Category: Politics and Regulation

Whither the randomized controlled clinical trial?
With the rise of precision medicine and genomics, the conventional randomized clinical trial appears more and more outdated. Fortunately, clinical trials are evolving, but will it be enough to incorporate the numerous advances in "-omic" medicine in a rigorous scientific manner to benefit patients?

Is there a reproducibility “crisis” in biomedical science? No, but there is a reproducibility problem
Reproducibility is the key to scientific advancement. It has been claimed that we suffer from a "reproducibility crisis," but in reality it is a chronic problem in reproducibility. Here we will look at the scope of the problem and strategies to address it.
Supplements, Lies, and a Lengthy Transcript
On October 21, 1993, there was a hearing before the U.S. Senate Committee for Labor and Human Resources, with the long-winded title: Examining How the Federal Government Should Regulate the Marketing and Use of Dietary Supplements and Related Measures, Including S. 784, To Strengthen Federal Standards with Respect To Dietary Supplements. S. 784, sponsored by Sen. Orrin Hatch, would eventually be enacted...
Change.org Petition: “Naturopaths are not physicians: stop legitimizing pseudoscience”
Britt Hermes, a graduate of the naturopathic college at the alternative medicine-focused Bastyr University, renounced her practice as a naturopathic doctor when she could no longer tolerate the pseudoscience and patient harm that characterizes naturopathy. On this blog and her own, Naturopathic Diaries, she has chronicled the insufficient education and training students receive before being allowed to practice as naturopathic doctors, deficiencies...

Nobody licenses quacks in my state! HB 4531 and the licensing of naturopaths in Michigan
Over the years, I’ve taken care of women with locally advanced breast cancer so advanced that it’s eroded through the skin, forming huge, nasty ulcers filled with stinky dead cancer tissue that’s outgrown its blood supply, leaving the patient in chronic pain. If the patient is fortunate, her cancer has not metastasized beyond her axillary lymph nodes (the lymph nodes under her...
Legislative Alchemy: Michigan House Bill 4531 gives naturopaths a broad scope of practice
Michigan House Bill 4531, if passed, would give naturopaths one of the broadest scopes of practice in the U.S., essentially equaling that of a family practice MD or DO. The bill made it through all the necessary House committees and is now before the House for an initial vote determining whether it will proceed further in that body. If it passes there,...
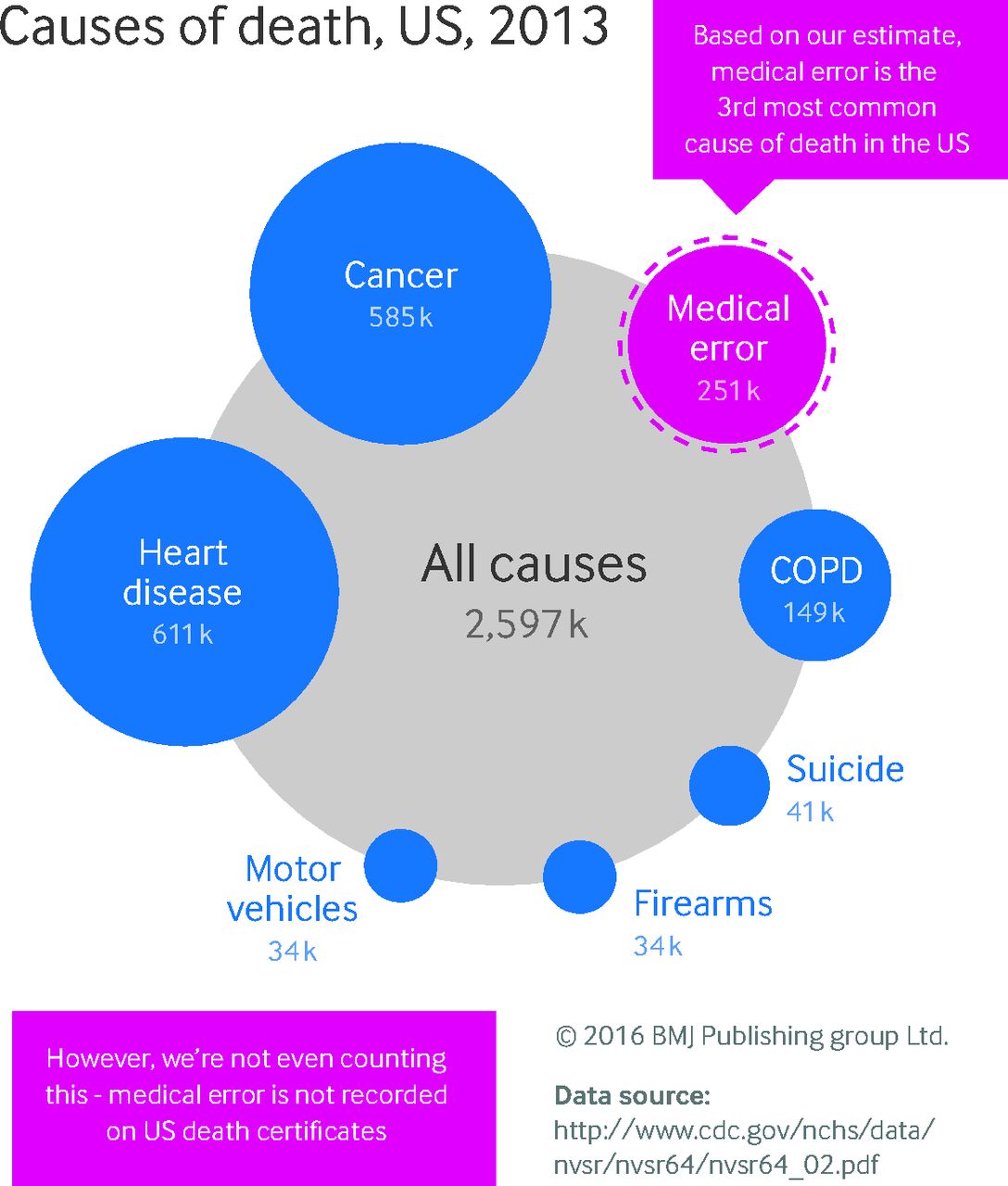
Are medical errors really the third most common cause of death in the U.S.?
A regurgitation of existing data suggested that medical error is the third leading cause of death in America. Is it true? Spoiler alert! No. No it's not. While medical error can and should be reduced, this BMJ article does not justify claims that doctors are a leading cause of death in the United States.

Medical exemptions to vaccine mandates for sale after SB277! Get ’em before they’re gone!
When California passed SB 277 into law, eliminating personal belief exemptions to school vaccine requirements and permitting only personal medical exemptions, I predicted that antivaccine quacks would start issuing bogus medical exemptions. Unfortunately, I was right.
What (if anything) does “natural” mean?
What does the term “natural” mean on a label? Does it mean anything? Should it mean anything? Good questions. And complicated ones, judging from the list of questions the FDA needs your help in answering. The FDA has resisted defining “natural” in food product labeling, including whether foods that are genetically engineered, or contain genetically engineered ingredients, can use the term. Back...
Reclassifying thyroid cancer and the willful misunderstanding of overdiagnosis
If there’s one lesson that we here at Science-Based Medicine like to emphasize, it’s that practicing medicine and surgery is complicated. Part of the reason that it’s complicated is that for many diseases our understanding is incomplete, meaning that physicians have to apply existing science to their treatment as well as they can. The biology of cancer, in particular, can be vexing....

