Category: Science and Medicine

Vaginal Seeding: To Swab or Not to Swab Your Newborn Baby
More mothers are requesting vaginal seeding, but does the evidence support this potentially risky intervention?
Do anti-inflammatory drugs effectively treat spinal pain?
While anti-inflammatory drugs are commonly used to treat back pain, a new review suggests that they may not provide meaningful benefits to most people.
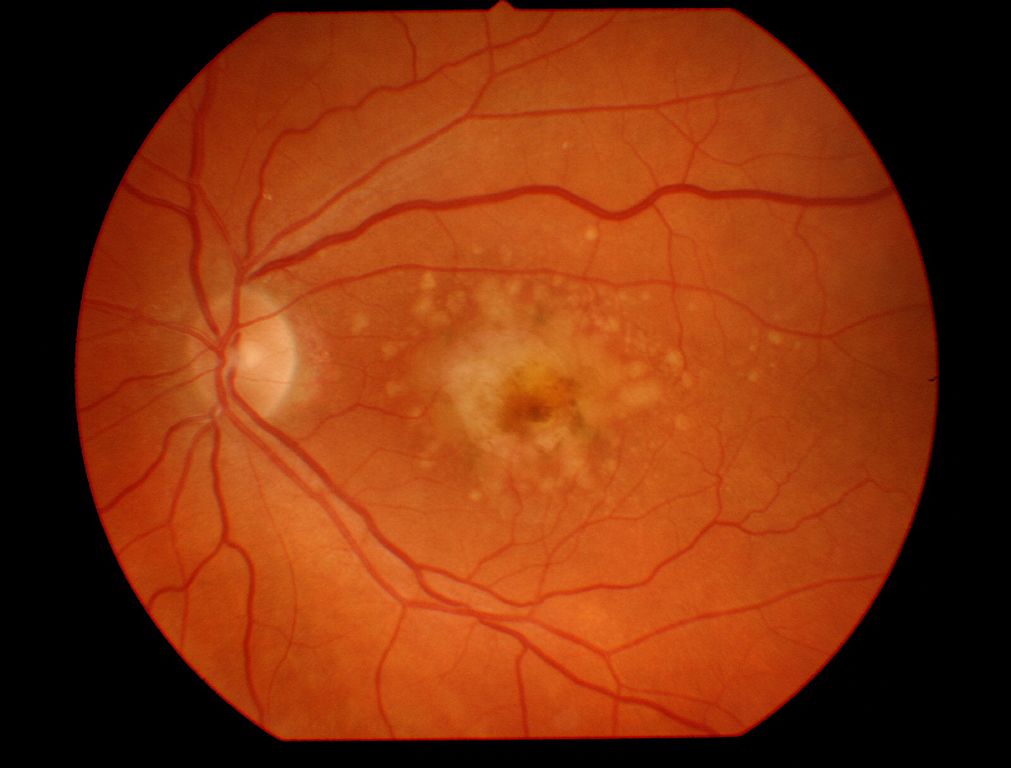
Re-thinking Antioxidant Supplementation for Macular Degeneration
After the AREDS trial, people with moderate to severe age-related macular degeneration were advised to take dietary supplements to slow the progression of the disease. But some experts say the trial actually showed supplements don't work, and might even make some patients worse.
Corrigendum: The week in review for 2/5/2017
Drinking hydrogen peroxide kills. Homeopaths don't care if their nostrums kill children. Acupuncture is placebo. But you knew that. Saudi Arabia bans reiki for the damnedest reasons. Eating placentas. And more!

Elemonics – Nothing but a dance and a tune
When it comes to quackery and pseudoscience, Mike Adams of NaturalNews.com has few peers. Amusingly, he thinks he's a scientist, too, and he's at it again.

Corrigendum. The Week in SBM for 01.29.2017
Not every article and study that pops up my feeds in the world of pseudo-medicine is worthy of a complete blog post. But they need to be noticed and commented upon: FDA confirms elevated levels of belladonna in certain homeopathic teething products. Homeopaths prove water not toxic to fish. Lots of acupuncture recommendations, little good data. Everything is CAM. And more! Duty...
Acupuncture for Infant Colic Part 2: Acupuncture Boogaloo
A detailed discussion of infant colic plus a few more thoughts on why acupuncture does not play a role in science-based management.

Cleveland Clinic Fully Embraces Pseudoscience
A recent and embarrassing anti-vaccine screed from the Director of the Cleveland Clinic Wellness Center produced a media backlash. Toby Cosgrove, CEO and President of the Cleveland Clinic, had the opportunity to re-dedicate his organization to good science and medical practice. Instead he doubled-down on the Cleveland Clinic's embrace of quackademic medicine and pseudoscience.

Peanut Allergy Prevention Advice Does a 180
New guidelines suggest that preventing peanut allergies may be as simple as giving peanut-containing food, beginning in infancy. How did old guidelines, which recommended avoidance, get it so wrong?

Newborn Chiropractic, False Balance, and The Doctors
The vacuous TV docs on The Doctors have demonstrated once again why the show is a highly unreliable source for medical information of any sort.

