The Ethics of Involuntary Pediatric Drug Testing
Although it may seem like a good idea, testing for recreational drug use on an adolescent patient without consent is ethically questionable, challenging to interpret, and unlikely to benefit patient or family.
A closer look at penicillin allergies
While many people believe they may be allergic to penicillin, few actually are. The consequences may be serious.
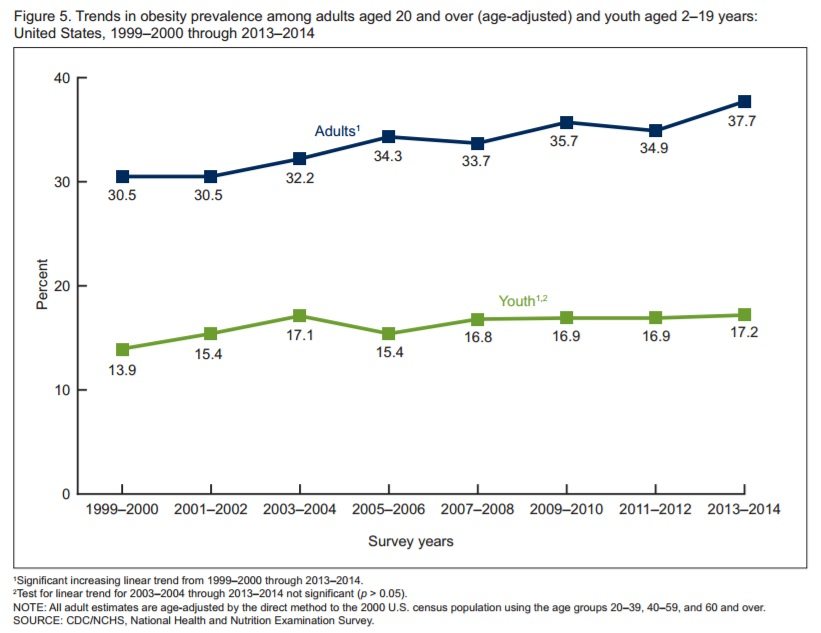
BMI and the Obesity Epidemic
Obesity is a serious and growing health problem worldwide. Ditching BMI as a measure of obesity is not the solution.

On Guard, DōTERRA, Essential Oils, and a Lesson in Reading Research Studies
A study of On Guard™, a mixture of essential oils, showed that it reduced the infectivity of influenza virus in dog kidney cells in the lab; but that's irrelevant to the question of whether the product has any clinical effect in humans.

Does society try to shame and shun vaccine refusers and the vaccine-averse?
Antivaxers often complain that they are judged harshly, even shunned. A recent study suggests that, to some extent, they might well be. But are judging, shaming, and shunning parents who refuse to vaccinate their children wrong? More importantly, what about the children, who didn’t choose not to be vaccinated, and how likely is such stigmatization to change behavior?
Chiropractic Subluxation Theory: Legal Support for Inappropriate Use of Spinal Manipulation
Legally licensed, unbridled subluxation-based chiropractors who offer unproven treatment for a broad scope of health problems endanger public health, stigmatize appropriate use of spinal manipulation, and deter development of chiropractic as a legitimate back-care specialty.

What naturopaths really want
Naturopaths want the same scope of practice as MDs and DOs, allowing them to make important medico-legal judgments that put life, liberty and property at risk.
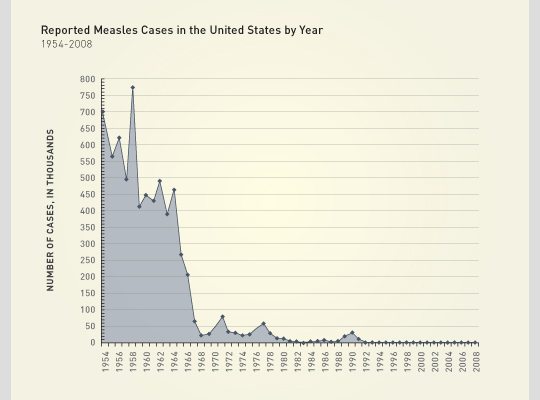
Minnesota Measles
There is currently an outbreak of measles in the Somali population of Minnesota – an outbreak that can be directly linked to antivaccine propaganda.
When Drugs Cost Too Much
Our ability to develop new drugs is fast outstripping our ability to pay for them; some are exorbitantly expensive and not very effective. Funds are limited, and as a society we need to have a serious discussion about how they are to be allocated.

The closure of major integrative medicine “Crown Jewels”: Terminating the Terminator?
When it comes to expansion and infiltrating medicine, "integrative medicine" has frequently seemed like the Terminator: utterly relentless. Recent setbacks at major integrative medicine "Crown Jewels" resulting in their closure cast that narrative in doubt. However, I never forget that after its seeming destruction, the Terminator always comes back.