Tag: Genetically modified organisms (GMOs)

Update on GMOs and Health
Thirty years on, there is even more evidence that GMO food are safe.
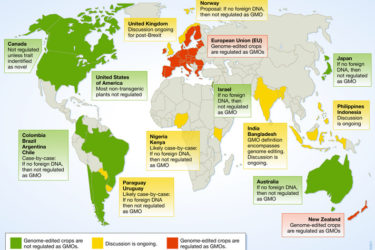
Gene Edited vs Genetically Modified
Why regulations should, and some are, distinguishing gene edited crops from GMOs.

Philippines Approves Golden Rice
Golden rice passes the final regulatory hurdle in the Philippines. Farming can now start planting.
Can GMOs cause gastrointestinal sensitivity and inflammation?
A closer look at the widespread claim that GMOs cause bowel irritation and inflammation.

