Tag: polio

Impressive New Data on the Benefit of Childhood Vaccines
A new study adds compelling evidence that vaccines given to young children prevents disease and saves the lives of people of all ages. It may not be surprising, but it's nice to see. Spread the word.

Can COVID Vaccines Travel in Space and Time?
Are COVID vaccines to blame for the return of polio?
Polio’s Resurgence
Surely we can keep iron lungs relegated to museums, can't we?
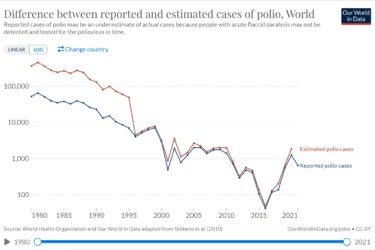
The Return of Polio
Once again we lose the chance to eradicate polio, but the goal still remains relatively close.
Deciding Which Risks to Take
No medical treatment is risk-free. Paul Offit's new book covers the history of innovations that went awry and advises how to balance the risks of new medical innovations with the risk of not treating.
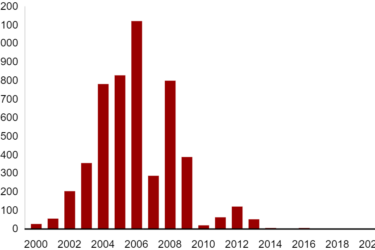
Polio Eliminated From Africa
Wild-type polio has been eliminated from Africa. Now only Pakistan and Afghanistan remain.
CDC Issues Warning: Be on the Lookout for a Surge in Pediatric Paralysis Cases This Fall
The CDC is warning parents and pediatric medical professionals to be on the lookout for a potentially devastating viral disease this fall. It isn't the novel coronavirus, but the impact of the pandemic might make things worse...or better.

Wrong About Polio: A Review of Suzanne Humphries, MD and Roman Bystrianyk’s “Dissolving Illusions” Part 1 (the long version)
This is a longer version of my post on Friday, November 9th, 2018. It is a lengthy discussion of why Suzanne Humphries, MD and Roman Bystrianyk's book Dissolving Illusions misrepresents the dangers of polio, one in a series of posts that should comprehensively show the problems with their claims. It covers far more than just polio, but is worthwhile for those interested...

Wrong About Polio: A Review of Suzanne Humphries, MD and Roman Bystrianyk’s “Dissolving Illusions” Part 1 (the short version)
Suzanne Humphries and Roman Bystrianyk's book Dissolving Illusions make numerous egregious errors or deceptive statements about polio. Here is part 1 in a series about the truth and true impacts of polio.
Polio Outbreak in Papua New Guinea
A recent case of polio on Papua New Guinea shows that we cannot rest until the eradication of polio is complete. Close is not good enough.

