Linus Pauling, the Nobel-prize winning researcher who slid into quackery shortly after.
Vitamin C as a treatment for cancer is back in the news again.
I’m not surprised. This is one therapy favored by advocates of “alternative” medicine that keeps popping up periodically (seemingly every couple of years or so). This latest bit of news has turned up almost right on time after the last time there was a push for rehabilitating vitamin C as a cancer cure a couple of years ago. Back in the spring of 2006, there were two studies published (more on them later) which were touted by the “complementary and alternative medicine” (CAM) crowd as evidence that Linus Pauling was supposedly vindicated. A little less than two weeks ago, an animal study was published suggesting that high-dose intravenous vitamin C had antitumor activity in mouse models. A couple of weeks prior, there had also been published a phase I clinical trial that showed that megadoses of IV ascorbate were safe and well-tolerated in cancer patients if they were appropriately screened for renal disease. Given the latest studies of this particular modality against cancer, it seemed like an opportune time for me to examine this new evidence and ask the question: Has Linus Pauling been vindicated?
I’ll cut to the chase. The short answer is: Not really, with the qualification that it depends on what you mean by “vindicated.” The long answer follows.
The Nobel disease
It’s been noted that there appears to be a tendency among Nobel Prize recipients in science to become enamored of strange ideas or even outright pseudoscience in their later years. Indeed, it’s happened often enough that some wags have dubbed this tendency the “Nobel disease.” Be it Linus Pauling and his obsession with vitamin C, Nikolaas Tinbergen and his adoption of the “refrigerator mother” hypothesis as the cause of autism (which has led one blogger going by the ‘nym Prometheus to quip that Tinbergen’s Nobel acceptance speech represented a “nearly unbeatable record for shortest time between receiving the Nobel Prize and saying something really stupid about a field in which the recipient had little experience”), or Louis J. Ignarro going from a Nobel Prize in Medicine for his work in figuring out nitric oxide signaling pathways to pushing his book on arginine supplementation as a cure-all for heart disease and becoming a shill for Herbalife, there’s something about becoming a Nobel Laureate that has a tendency to lead people to becoming cranks. Either that, or maybe it’s because mavericks who make Nobel-worthy discoveries have a tendency not always to recognize that not all of their ideas are as brilliant as the ones that garnered the Nobel Prize for them, although certainly another possibility is that winning the Nobel Prize tends to give some scientists an inflated sense of their own expertise in fields of science not related to the ones for which they won their Nobel Prize in the first place. Maybe it’s a bit of all of these.
Linus Pauling’s descent into pseudoscience
Be that as it may, the concept that megadoses of vitamin C can cure cancer has been around for decades now, ever since two-time Nobel Laureate Linus Pauling first proposed it. It began in 1972, when Ewan Cameron hypothesized that ascorbate could have anti-cancer action by inhibiting hyaluronidase and thereby preventing cancer spread after Pauling had first proposed that taking 1,000 mg of vitamin C daily can reduce the incidence of colds by 45% for most people. It wasn’t long before the two teamed up, and in 1976 Pauling and Dr. Ewan Cameron reported that a majority of 100 terminal cancer patients treated with 10,000 mg of vitamin C per day survived three to four times longer than patients who were not so treated.
Unfortunately, as experimental clinical protocols go, this study was a complete mess. Linus Pauling was not a clinician and had no experience in clinical trial design, and it really showed. Even as a restrospective analysis, the paper was a total embarrassment. There was no standardization, no good matching of controls by age, stage of cancer, or performance status; given the terrible design, there was clearly serious selection bias going on at a minimum. The study’s flaws, which were too numerous to mention, rendered its results essentially meaningless. If you want a quote from his original paper that shows this better than anything, here it is: “We believe that the ascorbate-treated patients represent a random selection of all the terminal patients in the hospital, even though no formal randomization process was used.” Suffice it to say that, in a clinical trial, it is not sufficient to “believe” that your groups were properly randomized and matched. You have to show it. Indeed, Dr. William D. DeWys, Chief of the Clinical Investigations Branch of the National Cancer Institute’s Cancer Therapy Program, pointed out that Pauling and Cameron failed at even a rudimentary effort to control for these variables:
Cameron’s patients began getting vitamin C when Cameron judged them “untreatable” and their subsequent survival was compared to that of the control patients from the time they had been labeled “untreatable.”
DeWys reasoned that if the two groups were comparable, the average time from the initial diagnosis to “untreatable” status should be similar for both groups. But they were not. He concluded that many of Cameron’s patients had been labeled untreatable earlier in the course of their disease and would therefore be expected to live longer. DeWys also noted that more than 20% of the patients in the control group had died within a few days of being labeled untreatable, whereas none of Cameron’s patients had died. This, too, suggested that Cameron’s patients had had less advanced disease when they were labeled untreatable.
The bottom line was that, for the above reasons and others, Pauling and Cameron’s study was shockingly bad. Undeterred, they published a follow-up study in 1978 that purported to confirm the findings of their 1976 study. It was little better. Pauling and Cameron used many of the same patients and selected a different control group, but the patients were still not matched for stage of cancer, age, or performance or nutritional status. It, too, was a shockingly poorly-designed study, even for a retrospective study.
Three decades later, I have to wonder how these studies saw print. It turns out that they were originally published in the Proceedings of the National Academy of Sciences, which is not a clinical journal. Not surprisingly, given his Nobel Prizes, Linus Pauling was a member of the National Academy of Sciences. What is not really known much outside the scientific community is that thirty years ago members of the NAS could contribute papers to PNAS as they see fit and in essence pick their reviewers. Indeed, until recently, the only way that non-members could have papers published in PNAS was if a member of the Academy agreed to submit their manuscript for them (known as “communicating” it), and, in fact, members were supposed to take the responsibility for having such papers reviewed before “communicating them” to PNAS. Thus, in essence a member of the Academy could get nearly anything he or she wished published in PNAS, whether written by him or herself or a friend. Normally, this ability has not been such a big problem for quality, because getting into the NAS is so incredibly difficult and only the most prestigious scientists are invited to join. Consequently, PNAS is still a highly prestigious journal with a high impact factor, and most of its papers are of high quality. Scientists know, however, that sometimes Academy members use it as a journal of last resort to publish some of their leftover findings. They also know that on occasion, when rare members fall for dubious science, as Pauling did, they can “communicate” their questionable findings and get them published in PNAS unless they’re so outrageously ridiculous that even the deferential editorial board can’t stomach publishing them. All they have to do is to find a couple of sympathetic colleagues to review their manuscripts and then submit them. What keeps the overall quality of most of the journal’s articles high is primarily the desire of members of the Academy not to sully their names by communicating papers that they consider to be poor quality science. These days, submission requirements for PNAS are more rigorous, whether that manuscript is submitted by the member or “communicated” to the journal for another investigator. Even so, getting a paper published in PNAS is quite easy for an Academy member and incredibly difficult for a non-member who does not have the connections that allow him to line up an Academy member willing to act as referee and thus is forced to submit his manuscript directly to the journal. These observations largely explain how Linus Pauling could submit such shoddy studies to PNAS and have them published. (Thus endeth my chance for ever getting a manuscript of mine published in PNAS. Probably.)
Eventually, three different randomized trials of high-dose vitamin C used to treat cancer were carried out by the Mayo Clinic. The first study was published in the New England Journal of Medicine in 1979, and Pauling was not pleased. He wrote a letter to the editor complaining that most patients had had extensive prior chemotherapy, which in his view meant that they were immunologically compromised. Given that his concept was that high-dose vitamin C somehow boosted the immune system, he thought it was an unfair trial, even though his first trial had pointed out that the patients undergoing vitamin C treatment had received standard therapy, including surgery, radiation, and chemotherapy as appropriate. This was followed by two other randomized clinical trials, which when coupled with the first trial, involved 367 patients and failed to find a benefit from high-dose oral vitamin C in cancer. Meanwhile, some studies were pointing out that high-dose ascorbate was not without adverse reactions, including diarrhea, and even renal failure after high-dose IV administration. Since then, among conventional oncologists at least, vitamin C as a therapy for cancer has been viewed as having no value.
It is highly doubtful that the concept that vitamin C might cure cancer and other diseases would ever have been taken as seriously as it was for as long as it has been had it not been championed by a scientific figure as towering as Linus Pauling. Unfortunately, in his zeal, Pauling popularized his ideas not primarily by publishing in scientific journals, but mostly by writing books, giving talks, and forming his own institute to do experiments designed to prove his ideas. Another sure sign of a zealot, Pauling couldn’t tolerate data that contradicted his belief in vitamin C. Indeed, when data from the experiments of a colleague at his institute, Arthur Robinson, suggested that vitamin C at the doses advocated by Pauling might actually increase the rate of tumor growth in an experimental model in mice:
Robinson, a former student and long-time associate of Pauling, helped found the institute and became its first president. According to an investigative report by James Lowell, Ph.D., in Nutrition Forum newsletter, Robinson’s own research led him to conclude in 1978 that the high-doses (5-10 grams per day) of vitamin C being recommended by Pauling might actually promote some types of cancer in mice [18]. Robinson told Lowell, for example, that animals fed quantities equivalent to Pauling’s recommendations contracted skin cancer almost twice as frequently as the control group and that only doses of vitamin C that were nearly lethal had any protective effect. Shortly after reporting this to Pauling, Robinson was asked to resign from the institute, his experimental animals were killed, his scientific data were impounded, and some of the previous research results were destroyed. Pauling also declared publicly that Robinson’s research was “amateurish” and inadequate. Robinson responded by suing the Institute and its trustees. In 1983, the suit was settled out of court for $575,000. In an interview quoted in Nature, Pauling said that the settlement “represented no more than compensation for loss of office and the cost of Robinson’s legal fees.” However, the court-approved agreement stated that $425,000 of the settlement was for slander and libel. The Institute’s own legal fees were close to a million dollars [21].
In 1994, Robinson and two colleagues summarized the results of four mouse studies he had carried out while working at the Pauling Institute. Nearly all of the mice developed skin cancers (squamous cell carcinomas) following exposure to ultraviolet radiation. Altogether, 1,846 hairless mice received a total of 38 different diets. The researchers found that (a) the rate of onset and severity of tumors could be varied as much as 20-fold by just modifying dietary balance; (b) diets with the worst balance of nutrients had the greatest inhibitory effect on cancer growth; and (c) no cures or remissions were observed (although the researchers were not looking for this). In 1999, Robinson commented:
The results of these experiments caused an argument between Linus and me, which ended our 16-year period of work together. He was not willing to accept the experimentally proved fact that vitamin C in ordinary doses accelerated the growth rate of squamous cell carcinoma in these mice.
At the time, Linus was promoting his claim that “75% of all cancer can be prevented and cured by vitamin C alone.” This claim proved to be without experimental foundation and not true. . . . Vitamin C increased the rate of growth of cancer at human equivalents of 1 to 5 grams per day, but suppressed the cancer growth rate at doses on the order of 100 grams per day (near the lethal dose), as do other measures of malnutrition [23].
Of course, this could just be sour grapes after a falling out among old friends, or it could be only one side of a more nuanced story. It could also be that the acceleration of growth reported by Robinson may not be the behavior of all tumors or even most tumors in response to high-dose vitamin C. However, if Robinson’s results as he reported later were correct, in the case of at least one tumor vitamin C appeared not to inhibit growth but rather to accelerate it. Pauling’s apparent willingness to suppress data that did not fit with his vitamin C hypothesis and to fire the man whose experiments yielded such data must be acknowledged, particularly given the libel settlement that resulted from legal action by Robinson. The representation of Pauling as a scientist unfairly vilified for his ideas that were outside the mainstream may have an element of truth to it, but just an element. The whole story is not nearly so flattering to Pauling, and he deserved more than a little of the vilification heaped upon him for his vitamin C advocacy and for his role in the promotion of orthomolecular medicine, which consisted mostly of recommending megadoses of vitamins and nutrients to treat disease.
Of course, falling from grace as Pauling did led to wishes for that most American of stories: His triumph and vindication, even if posthumous. At least, so Pauling’s admirers have been hoping for at least 20 years.
The 21st century “vindication” of Linus Pauling? (Take 1)
One aspect of dubious “alternative” therapies is that virtually no amount of evidence can persuade their adherents that their favored therapy really and truly does not work. However, in the case of vitamin C as a cancer therapy, one loophole in the clinical studies that did not show efficacy is that in all of well-designed randomized clinical trials, it was oral vitamin C that was studied. The evidence regarding high-dose intravenous dosing was not as copious, and this oversight was exploited by all manner of CAM practitioners, some of whom promised near miraculous results with intravenous vitamin C, sometimes combined with various other supplements and minerals, while even as early as 2000 some “conventional” physicians and scientists suggested that a reevaluation was in order or that intravenous vitamin C could improve the quality of life for cancer patients.
It turns out that emerging data showed that ascorbate was differentially toxic to some tumor cell lines at concentration in the millimolar to tens of millimolar range. That’s an incredibly high concentration to have to achieve in blood and tissues. Indeed, if you told most pharmacologists that you had a new experimental compound for the treatment of cancer (or any other disease, for that matter) but that it required blood and tissue concentrations of 10 mM to be effective, nearly all of them would suggest that you give up in a hurry because of how difficult it is to reach concentrations that high in humans without toxicity. Even so, two and a half years ago, there came a study published in PNAS by Mark Levine and colleagues at the National Institute of Diabetes and Digestive and Kidney Diseases at the National Institutes of Health. The PNAS study was simply an in vitro study using five tumor cell lines that showed selective toxicity due to ascorbate in tumor cells compared to normal cells at concentrations that are achievable with intravenous doses of ascorbate. It turns out that ascorbate was four- to twenty-fold more toxic to the tumor cells tested than to normal cells, and that cell killing depended upon hydrogen peroxide generation, as it was abrogated when cells were pretreated with peroxide scavengers. It’s a pretty straightforward paper. My only quibbles with it were that the concentrations of ascorbate used were very high, in the 2 to 20 mM range (hence the need for very high intravenous doses of vitamin C to achieve such levels in the blood) and that Levine only used monocytes, lymphocytes, fibroblasts, and breast epithelial cells as the “normal” cells. Also, he only exposed the cells to the ascorbate for one hour, even though the paper itself states that it can take several hours for a large intravenous dose of vitamin C to be eliminated from the body. At the time I thought that the paper represented an interesting in vitro observation, but, as they say, you can kill tumor cells in vitro with just about anything if you crank the concentration high enough. In vitro experiments can generate hypotheses, but they are inherently highly artificial systems. As clinicians, we want to know if this works in vivo. Many are the chemotherapeutic agents that appear to kill cancer cells selectively in vitro but fail in animal experiments. All in all, this study was hardly a stunning vindication of Pauling.
At around the same time, another study was published in the Canadian Medical Association Journal (CMAJ), also by Mark Levine and colleagues in collaboration with a group at McGill University. Based on the concept that only intravenous dosing can produce high enough blood levels of ascorbate and that that is the reason previous trials failed to show a benefit from high-dose vitamin C, three cancer cases were reported, all of whom received high-dose vitamin C and all of whom, it was claimed, showed much longer-than-expected survival. The problem is, there was a lot less there than has been reported, especially given that this study was of a type that I particularly detest (but that CAM advocates particularly love), the so-called “best case” series.
The first patient of the three reported, for instance, had renal cell carcinoma with putative lung metastases and received 65 g vitamin C intravenously twice a week for 10 months. The lung metastases were never biopsy-proven to be renal cell carcinoma, leaving some doubt about them. However, let’s accept that they probably were real lung metastases, even though none of them were ever biopsied. (We don’t always biopsy metastases to prove that they are metastases, particularly if the biopsy would be unduly risky. However, in oncology generally it is always better to have a tissue diagnosis than not.) The problem is that renal cell carcinoma is a tumor type that is one of the more common tumor types to be reported to undergo unexplained spontaneous remissions. It’s uncommon, but it has definitely been documented. Also, the patient received a number of other “alternative therapies,” including thymus protein extract, N-acetylcysteine, niacinamide, and whole thyroid extract. This led me to ask: If the authors believed that alternative therapies like high-dose vitamin C can cure certain cancers, why did they automatically assume that it had to be the vitamin C? Why couldn’t it have just as plausibly been one of the other treatments or some combination of them? The fate of this patient was ultimately not good: “Unfortunately, the woman — a long-time smoker — was diagnosed four years later with small-cell lung cancer. She again opted for vitamin C treatment, but the second cancer did not respond.” Was there vitamin-resistance in the second tumor as opposed to the first, or did the first tumor just go into spontaneous remission, or was it something else? It’s impossible to say much of anything from this case.
The second case presented was of a man who had a primary bladder tumor with multiple satellite tumors. These were all resected with apparently clear margins transurethrally using a cystoscope. Rather than additional conventional therapy, the man chose intravenous vitamin C and was reported alive and well nine years later. Normally, this would be a pretty poor prognosis tumor without more therapy, but, as the accompanying commentary points out, long term survival has been reported with surgical therapy alone. Indeed, this case reminds me a lot of breast cancer patients who undergo excision of their primary tumor, forgo adjuvant therapy for alternative therapy, and then attribute their survival not to the conventional therapy (surgery) but rather to the alternative therapy. In addition, this patient also took a variety of other alternative therapies, leading to the question again: Why did the authors assume that it was the vitamin C? They may be correct, but there are too many confounding factors. It could just as plausibly been the surgery.
Finally, the third patient had a large paraspinous mass that turned out to be diffuse B-cell lymphoma. She agreed to radiation therapy but refused chemotherapy, opting instead for intravenous vitamin C. She, too, used a variety of other alternative therapies, including beta-carotene, bioflavonoids, chondroitin sulfate, coenzyme Q10, dehydroepiandrosterone, a multiple vitamin supplement, N-acetylcysteine, a botanical supplement, and bismuth tablets. It’s utterly ridiculous to include this case as “evidence” for the efficacy of vitamin C. Besides the confounding factor of using multiple alternative therapies, the patient reported only had, as far as can be told from the case report, a Stage I diffuse B cell lymphoma. Radiation therapy alone used to be a common treatment for such tumors and is well known to be able to produce long term survival in such cases. To try to show that this was something other than Stage I disease, the authors made much of the observation that the patient subsequently developed enlarged lymph nodes in numerous other nodal basins over ten years. However, nowhere did they report that any of these enlarged lymph nodes were ever biopsied to prove conclusively that they were recurrent lymphoma, and the only pathology result reported was from the biopsy of the paraspinous mass. One thing oncologists that I’ve worked with damned near always insist upon before diagnosing a recurrence of lymphoma is proof by tissue diagnosis, and it is very puzzling why such a simple procedure was not done at some point over the ten year case history reported. It would have been very easy to do biopsies of any of the superficial lymph nodes that were reported to have become enlarged. Another point I noticed was that, looking at the authors’ descriptions, only one of these nodes would I have even considered particularly suspicious (a 3 cm supraclavicular node). The majority of the nodes reported were merely described as merely “palpable” or as measuring 1.0 or 1.5 cm, which is within the normal range for lymph nodes in those basins.
This study is one reason why I really, really detest “best case” series. CAM advocates love such series, though, because they allow them to cherry-pick patients. The problem is this: Even if these three cases do represent legitimate tumor responses to vitamin C, we have no idea what the denominator is. These cases were collected over many years, and it’s impossible to know how many thousands of patients underwent similar vitamin C regimens with no response at all. In fact, I find it hard not to point out that these were the very best cases the authors could come up with over a span of many years, and yet they are only marginally more convincing than the anecdotes. The bottom line: as of 2006, the evidence supporting the use of vitamin C in cancer, either intravenous or oral, was not compelling. Considering that even in theory very high ascorbate concentrations would be needed, meaning very high-doses, for an effect that in vitro was fairly weak, made the whole thing seem like a long run for a short slide.
The 21st century “vindication” of Linus Pauling? (Take 2)
Fast forward to July and August 2008.
Two and a half years have passed, and Levine et al are back, this time with another study published in PNAS entitled “Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice.” This time, they’ve done animal studies. Once again, these studies show mildly positive results. Once again, the whole obsession with using vitamin C to treat cancer, even if Levine’s results hold up as reported, strikes me as a long run for a short slide. Once again, I remain puzzled as to why there is such intense interest in what, even if it “works,” strikes me as a cumbersome and only mildly effective anticancer therapy.
How good is the study? Answer: It’s OK but it’s nothing special. In fact, it’s not particularly “meaty,” with only four figures, one of which is essentially a rehash from Levine’s previous PNAS paper with some extra cell lines added. Indeed, if this paper were not about a controversial and questionable therapy that had been thought to have been found wanting and instead been about a new cancer chemotherapeutic, my guess is that this result would have been viewed as mildly promising but nothing earthshaking. However, it’s not about some new chemotherapy agent; it’s about vitamin C. Consequently, it got a fair amount of attention in the press when it came out a couple of weeks ago.
Basically, this study reports the results of in vivo experiments in mouse tumor models using high-dose vitamin C to treat tumors. In the introduction, Levine argues that the reason previous studies using high-dose vitamin C to treat cancer failed to show an antitumor effect was because the drug was administered orally. The human body, he points out, keeps a pretty tight control over the allowable blood concentration of vitamin C, and it’s not possible to pump that concentration up above a certain level through oral dosing alone. If the blood level of ascorbate could be boosted far beyond the limit that can be achieved with oral dosing, Levine hypothesized, it might reach concentrations in the 10 mM range, which is the concentration range in which he has shown that ascorbate can kill tumor cells in vitro.
One very important thing to emphasize here, though, is that at these doses ascorbate is not acting as a nutrient. It is not a “nutritional” treatment for cancer, as some CAM advocates claim. Rather, it is acting as a drug, more specifically a prodrug that generates peroxide and ascorbate radicals. (It is not an uncommon for antioxidants to become prooxidants when administered at high-doses.) Indeed, these are doses that are many, many, many times greater than what is required for good nutrition; they are true “megadoses.” To illustrate the point, I note that the doses used in mice by Levine et al were on the order of up to 4 g/kg twice daily. Translating that dose to the proverbial 70 kg man would mean a dose of up to 560 g per day, or nearly a pound and a quarter, administered intravenously. Of course, mice are not humans, and, as it turns out, it doesn’t take doses that high to reach blood concentrations in humans as high as those achieved in the mice using 4 g/kg ascorbate; it takes up to 1.5 g/kg (more on that later). Even so, we’re still talking huge doses of ascorbate just to get its blood and tissue concentrations into a theoretical potentially therapeutic range. Given that a typical orange contains approximately 45 mg of vitamin C, 1.5 g/kg administered to a 68 kg adult would translate into over 2,000 oranges’ worth of vitamin C. It’s also a ten-fold higher dose than was used by Pauling and Cameron.
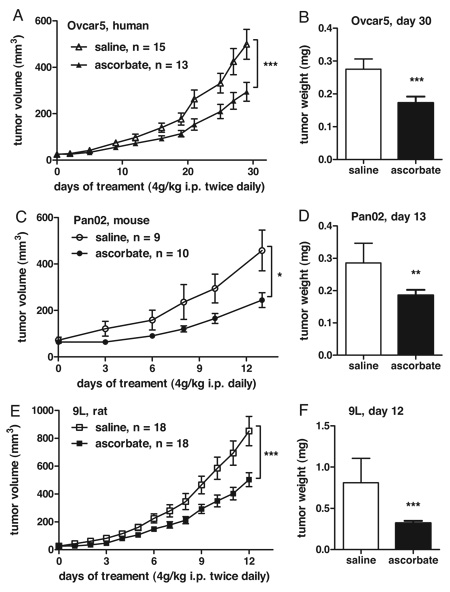
After in essence recapping results presented in Figure 1 of their previous study for this study again and adding more cell lines to the panel to show that high concentrations of ascorbate are selectively toxic to tumor cells compared to normal cells, Levine gets to the meat of his results by testing his vitamin C regimen in three different mouse models of cancer in athymic nude mice: Ovcar5, a human ovarian cancer cell line; Pan02, a mouse pancreatic cancer cell line; and 9L, a rat glioblastoma cell line. Why he chose these particular cell lines is not really explained clearly. Personally, if I were doing this experiment I would have made sure to use nothing but human tumor cell lines; in this study only one of the xenograft models used was human. Be that as it may, tumors were allowed to grow to a volume of 50 ± 10 mm3, at which point treatment with either intraperitoneal injections of ascorbate or saline placebo were commenced. The results are below:
As you can see, the growth of the tumors was delayed, but there doesn’t appear to be any tumor shrinkage. It turns out that this is actually not a particularly uncommon result for a potential cancer chemotherapeutic drug (and, make no mistake, in this study ascorbate is being used as a chemotherapeutic agent). That’s why I don’t hold it against this study. I agree, based on the data presented, that against these three cell lines, at least, ascorbate at these doses does appear to exhibit some in vivo antitumor activity. However, it’s modest and it requires massive doses producing high concentrations of ascorbate in the blood to achieve, but it’s clearly there. At the end of the study, treated tumors were anywhere from 40-53% smaller than the untreated controls, and in the 9L cell line there was reportedly a major decrease in the frequency of detectable metastases. That’s fairly convincing. The rest of the study involved mainly measuring the levels of H2O2 in the tissues of treated animals. Amazingly, peak levels of as high as 30 mM were measured, and high concentrations of the oxidant ascorbate radical and H2O2 were measured.
The observation that such high ascorbate concentrations in the mice’s blood could be achieved led the authors to reference a phase I clinical trial of high-dose intravenous vitamin C in advanced malignancy that had been published two weeks earlier and reported that it is possible to approach such high concentrations of ascorbate in humans with doses of 1.5 g/kg. Even though that’s not 4 g/kg, that dose is still a lot and still problematic, as the infusion of high volumes of high osmolarity solution did cause problems in this trial. Nonetheless, according to this phase I study, with appropriate screening for conditions that might predispose to adverse events, especially renal failure, the results of the phase I clinical trial suggest that high-dose intravenous vitamin C is fairly well tolerated even at the highest dose range.
Based on the animal studies and the safety data from the phase I trial, Levine concludes that high-dose intravenous ascorbate represents a promising new therapy that should be further investigated. If his were the only evidence I saw being presented, I’d agree that it’s mildly to moderately promising as an anticancer therapy. However, I find Levine’s results much less compelling in light of the history of the use of vitamin C in cancer and in light of my knowledge of cancer chemotherapeutics. More importantly, I know that the phase I clinical trial described above did not identify even a single patient with an objective response to therapy. In other words, not one patient’s tumor shrank. True, the lack of a single objective response in a phase I trial does not necessarily mean that a new cancer chemotherapy drug doesn’t work, but it certainly does dampen considerably any optimism that that the drug will work as a single agent. Taking the animal studies and the phase I trial together, I view the results with mild optimism that high-dose vitamin C might have efficacy in human cancer and be useful in combination with other chemotherapy. However, putting these results in context with what has been reported before over 30 years decreases even that mild enthusiasm, especially when coupled with my observation of just how modest the responses were in the mice and by just how much ascorbate was required to result in even a mild antitumor effect. Indeed, the authors of the phase I trial even admitted:
In summary, this study shows that 1.5 g/kg ascorbic acid infused >90-120 min three times weekly is essentially free of risk and important side-effects when simple precautions are taken. In people with normal renal function, this dose achieves a plasma ascorbic acid concentration >10 mmol/l for several hours. No patient experienced an objective anticancer response, although two patients at the 0.6-g/kg dose received greater than six cycles of ascorbic acid with stable disease. Even though only six patients received the recommended phase II dose, our results suggest that the likelihood of an objective anticancer response to i.v. ascorbic acid alone is slight in unselected patients with multiply treated advanced cancer.
I do note the qualifiers in this admission: unselected patients and multiply-treated advanced cancer. The latter qualification is in essence meaningless, because no one is going to use an agent like vitamin C as a first line therapy for any cancer if there is not strong clinical trial evidence showing that it works at least as well as current chemotherapeutics. To do otherwise would be highly unethical. On more solid ground is the former qualifier, as there conceivably might be a subpopulation that will respond to high-dose vitamin C. Even if that is true, however, based on the results of this phase I trial and the animal studies, clearly vitamin C is not any sort of “miracle cure.”
Linus Pauling: Vindicated or not?
All of this brings us back to the title of this post, to the question that just can’t be avoided: Do these two studies released in the last month vindicate Linus Pauling and all the alternative practitioners of high-dose vitamin C therapy for malignancy? Again, it depends on what you mean by “vindicate,” but my answer is nonetheless: Not really, except perhaps in the weakest of ways.
Here’s why. Linus Pauling didn’t claim that vitamin C in combination with other chemotherapeutic drugs might provide an additional incremental benefit to patients, which is the very best that can be claimed for high-dose ascorbate based on all the in vitro, in vivo, and clinical evidence thus far. That’s far too modest a claim. No, actually Pauling said that “75% of all cancer can be prevented and cured by vitamin C alone.” Similarly, boosters of vitamin C as a cancer therapy don’t claim that it provides a small additional benefit when administered with chemotherapy, either, which again is the very best that can be possibly claimed based on the experimental results described. Rather, they claim that high-dose vitamin C cures cancer a high percentage of the time. Even the most optimistic appraisal of Levine’s mouse study or the phase I clinical trial do not support any such claims. That much is brutally clear.
The bottom line from my perspective: Yes, vitamin C probably has some antitumor activity for some tumors, but as I contemplate the evidence for this effect the word “underwhelming” comes to mind, as does one of my favorite metaphors, namely the long run for a short slide. Apparently there are a lot of people who like getting their butts dirty sliding into base after a long run, though, because the attraction to vitamin C still lives on despite unimpressive evidence. Naturally, the authors of the phase I trial suggest using vitamin C in combination with chemotherapy, which is a perfectly reasonable approach for drugs that don’t show any objective evidence of activity as a single agent. However, this will not be easy, given that the sheer mass and volume of ascorbate that must be administered and the very high concentrations that are required even in the best case scenario could easily interfere with other chemotherapeutic agents.
One thing is fairly clear from the data reported thus far, even Mark Levine’s data and his calls to “revisit” the question of vitamin C and cancer with an “open mind”: If high-dose intravenous ascorbate has antitumor activity in humans, that activity is almost certainly quite modest at best, and to achieve even such modest antitumor activity definitely requires incredibly high-doses of ascorbate. Once again, I point out that any other experimental drug requiring such high plasma concentrations and high-doses to achieve such a modest antitumor effect would probably garner very little interest from anyone, even if it were a potentially patentable product of big pharma. It’s possible that high-dose IV ascorbate might ultimately find its way into the armamentarium of science-based oncology, but it’s very unlikely ever to become a mainstay of treatment for any malignancy. It’s just too wimpy.
References
1. Chen, Q., Espey, M.G., Sun, A.Y., Pooput, C., Kirk, K.L., Krishna, M.C., Khosh, D.B., Drisko, J., Levine, M. (2008). From the Cover: Pharmacologic doses of ascorbate act as a prooxidant and decrease growth of aggressive tumor xenografts in mice. Proceedings of the National Academy of Sciences, 105(32), 11105-11109. DOI: 10.1073/pnas.0804226105
2. Hoffer, L.J., Levine, M., Assouline, S., Melnychuk, D., Padayatty, S.J., Rosadiuk, K., Rousseau, C., Robitaille, L., Miller, W.H. (2008). Phase I clinical trial of i.v. ascorbic acid in advanced malignancy. Annals of Oncology DOI: 10.1093/annonc/mdn377