The New York Times has been periodically running a series about the “40 years’ war” on cancer, with most articles by Gina Kolata. I’ve touched on this series before, liking some parts of it, while others not so much. In particular, I criticized an article one article that I thought to be so misguided about how the NIH grant system leads researchers to “play it safe” and how we could cure cancer if we could just fund “riskier” research that I had to write an extended screed about the misconceptions in the article. The latest installment, Medicines to Deter Some Cancers Are Not Taken, also by Kolata, is much better in that it discusses a problem at the heart of cancer, namely that we have developed drugs that can decrease the risk of specific cancers but they are not as widely used as they could be.
The first part of the article contrasts a seeming incongruity:
Many Americans do not think twice about taking medicines to prevent heart disease and stroke. But cancer is different. Much of what Americans do in the name of warding off cancer has not been shown to matter, and some things are actually harmful. Yet the few medicines proved to deter cancer are widely ignored.
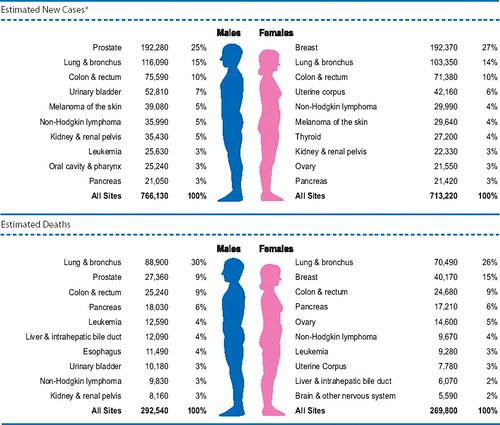
Take prostate cancer, the second-most commonly diagnosed cancer in the United States, surpassed only by easily treated skin cancers. More than 192,000 cases of it will be diagnosed this year, and more than 27,000 men will die from it.
And, it turns out, there is a way to prevent many cases of prostate cancer. A large and rigorous study found that a generic drug, finasteride, costing about $2 a day, could prevent as many as 50,000 cases each year. Another study found that finasteride’s close cousin, dutasteride, about $3.50 a day, has the same effect.
This is indeed a contrast. Think about it. Millions of Americans take statins, for instance, to lower their cholesterol and thereby try to prevent the complications of elevated cholesterol, such as heart disease, vascular disease, and strokes. Yet, for at least two common cancers, there are proven effective drugs that will lower the risk of cancer considerably with a side effect profile at least as favorable as that of statins.
Of course, preventing cancer is not like preventing heart disease. “Cancer” is not a disease, but many diseases arising from many different organs with many different biological behaviors. There is no drug that can prevent “cancer.” There are, however, specific drugs that can lower the risk of specific cancers. So right there you have a big difference that could partially explain the reticence. Which cancers should be prevented? How many different drugs are you willing to take to prevent how many different cancers?
One thing that the article points out that is quite true and has been quite disappointing to cancer researchers, and that’s that diet does not have nearly as large an effect as we had hoped. Kolata correctly points out that if we could eliminate smoking cancer deaths would decrease by about a third. Lung cancer is quite rare in nonsmokers. Indeed, back in the early part of the 20th century, lung cancer was so rare that there are reports describing how, when a patient died of it, medical students would be told that they had to go to see the autopsy because they might not ever see another case in their careers. Now lung cancer is the single largest cause of cancer deaths in both men and women, causing 30% of cancer deaths in men and 26% of cancer deaths in women, and it’s all due to smoking:
No other preventative intervention could conceivably even come close in terms of effect as cutting smoking to zero. Obviously, such a thing will not come to pass in my lifetime, but as a cancer surgeon I can always hope, can’t I? Also as a human being, too. After all, I’ve already lost an aunt to smoking-induced lung cancer in 2008, and I have several relatives who smoke who, I fear, might well join her.
After that, Kolata suggests that avoiding hormone replacement therapy containing estrogen and progestin after menopause would be a good preventative, which may be true, but since the Women’s Health Initiative study reported in 2002 that showed an increased risk of breast cancer from such HRT, few women take them any more anyway.
What’s more interesting is how Kolata correctly points out just how little diet and other lifestyle factors seem to matter compared to our hopes. If anyone wants to counter the claims of woo-meisters that we evil “allopathic” doctors don’t pay attention to diet and lifestyle, one only needs to look at just how much money has been spent on studies of diet and exercise and how they impact cancer and heart disease. There have been many studies. Of course, it’s incredibly difficult to do a randomized study, particularly a double-blinded one, of diet and exercise; so most of them end up being observational. The problem with such studies is, as with all such cohort studies, controlling for confounders. It’s hard enough to control for confounders that the investigators know about, but there are often many confounders that they don’t know about and may not identify until after the study is over. In any case, suffice it to say that studies of diet as a means of preventing cancer have been disappointing of late, as Kolata points out:
For example, public health experts for years recommended eating five servings of fruits and vegetables a day to prevent cancer, but the evidence is conflicting, at best suggestive, and far from definitive.
Low-fat diets were long thought to prevent breast cancer. But a large federal study randomizing women to a low-fat or normal diet and looking for an effect in breast cancer found nothing, said its director, Ross L. Prentice of the Fred Hutchinson Cancer Research Center in Seattle.
Fiber, found in fruits, vegetables and grains, is often thought to prevent colon cancer, even though two large studies found no effect.
“We thought we would show relationships that were strong and true,” said Dr. Tim Byers, professor of epidemiology at the Colorado School of Public Health, “particularly for dietary choices and food and vegetable intake. Now we have settled into thinking they are important but it’s not like saying you can cut your risk in half or three-quarters.” Others wonder whether even such qualified support is misplaced.
There has to be a reason the research disappointed, said Colin B. Begg, chairman of the department of epidemiology and biostatistics at Memorial Sloan-Kettering Cancer Center. Perhaps the crucial time to intervene is early in life.
“That’s one possibility,” Dr. Begg said. “The other is that it’s all sort of nonsense to begin with.”
One potential reason for these results is that the effect of diet or other environmental exposures may have a “window of vulnerability” that occurs in youth or childhood. Certainly, in the breast cancer field, we are starting to think that and actively research the possibility. The problem is, if that’s true, then dietary interventions during adulthood may have little or no effect on ultimate cancer risk.
Which brings us to drugs and supplements.
Kolata mentions the cautionary tales of beta-carotene and selenium, the former of which was thought to prevent cancer and the latter to prevent prostate cancer. However, when a large study of beta carotene was done, not only did the supplement not prevent cancer but it appeared to increase the risk of lung cancer in smokers. Selenium and vitamin E similarly were found to have no protective impact on prostate cancer risk and, in fact, the trial studying the question was halted because there was a hint of actually increased risk. Even so, as Kolata points out, ads for such supplements continue to imply, with the Quack Miranda warning, that such supplements “improve prostate health,” whatever that means, although the implication is that they decrease the risk of prostate cancer.
Now here’s where the drugs come in. Because I’m a breast cancer surgeon, I’m going to focus mainly on strategies to prevent breast cancer. It turns out that tamoxifen can decrease the risk of breast cancer by 50% in high risk women. Tamoxifen is a selective estrogen receptor modulator (SERM) that antagonizes estrogen action in breast tissues but has partial agonist activity in bone and endometrium. Indeed, raloxifene, another SERM, is used primarily to prevent and treat bone loss in women with osteoporosis. A $110 million clinical trial showed that its ability to prevent breast cancer is not statistically significantly different than that of tamoxifen, with the advantage that, unlike tamoxifen, raloxifene does not increase the risk of uterine cancer and does not increase the risk of blood clots as much as tamoxifen does.
So, with such overwhelming evidence that in women at high risk for breast cancer, why is it that so few are offered the option of tamoxifen or raloxifene? Kolata explains:
“Those were your tax dollars and mine,” he [Dr. Victor Vogel] added. “You can’t do too many $110 million studies.”
He cannot understand why no one cares, but some doctors say they see a number of problems. It is usually not the cost; tamoxifen is about 30 cents a day and raloxifene $3.30 a day. It is doctors’ practices and women’s concerns.
Most doctors, said Dr. Therese B. Bevers, medical director of the Cancer Prevention Center at M. D. Anderson, do not take the first step — calculating a woman’s lifetime risk of getting breast cancer — in part because that can lead to the next step, spending an hour or so discussing cancer risk and drug risks and benefits.
Dr. Bevers suggests the drugs for women whose lifetime odds exceeds 20 percent. That could include, for example, a 55-year-old woman who began menstruating early (increasing the risk), had her first child late (again increasing the risk), and whose mother and sister got breast cancer. About half the time, though, women with that kind of risk turn down the drugs, Dr. Bevers said. “The No. 1 reason I hear is, ‘Oh, I just don’t like to take medications,’ ” she added.
Yet the same women will happily down supplements that woo-meisters like Dr. Mercola or Mike Adams will suggest to them as cancer preventing. Some will down many supplements a day without thinking twice about it, even though there is no good evidence that they do anything other than enrich the supplement manufacturers who make them and they often cost far more than the paltry 30 cents a day that tamoxifen costs. Moreover, in general for prevention the usual course of tamoxifen or raloxifene is five years, in marked contrast to supplements, where the woo-meisters recommend them for the rest of one’s life.
Of course, never having worked anywhere else other than NCI-designated comprehensive cancer centers, I am probably spoiled in this respect. Such centers always have genetic counseling and prevention clinics. I can simply refer suitable patients there, where our trusty and excellent genetic counselors will calculate patients’ lifetime risk of breast cancer, construct family trees, decide if testing for cancer susceptibility genes is appropriate, and then make science-based recommendations for risk prevention strategies and/or screening strategies tailored to the individual patient. (Who says “allopathic medicine” doesn’t individualize therapy?) In any case, I can see why primary care doctors don’t bring these issues up more often, but with the advent of genetic counseling clinics I am puzzled why more patients aren’t referred to a specialist for these conversations.
One problem mentioned in the article is that there aren’t any particularly good biomarkers for increased risk for cancer, especially ones that fall in response to preventative interventions, the way, for example, cholesterol does for heart disease. Another is what I mentioned earlier, namely that cancer is not one disease, meaning that it is likely that many drugs would be needed to prevent various cancers. True, for the really common cancers, like prostate, breast, and colon, it might be worth it to develop such drugs, but neither the risk-benefit profile nor the number of patients who would benefit would be likely to lead to such drugs for less common cancers. And, as has been pointed out, our risk assessment tools are currently crude at best. In fact, even with better biomarkers I’m not so sure that interest would improve because biomarkers have their own problems and many people have a tendency to fear pharmaceuticals more than they fear a hypothetical increased risk of cancer. In the case of drugs that prevent breast cancer, nearly all of them have anti-estrogen activities and thus produce side effects of estrogen deprivation, such as hot flashes and other menopausal symptoms. It’s a very high bar to meet to produce effective drugs that people will actually take, given that it’s hard enough to persuade patients to comply with drugs designed to treat actual diseases and conditions.
The end result of all of this is that drug companies, once interested in developing cancer preventing drugs, are now floundering. As Kolata notes:
But risk assessment is not easy, and biomarkers are still more of a dream than a reality. There are other problems, too. If each cancer requires a different drug for prevention, how many drugs can a person take? For now, Dr. Curt said, the very idea of cancer prevention is daunting. And since cancer can take decades to develop, by the time a study concludes, a drug’s patent life may be over.
It is not a pretty picture, Dr. Vogel said.
“You have to think that in boardrooms they are saying, Man, did we learn a lesson,” he said. “We will stay as far away as possible from cancer prevention.”
Sadly, supplement manufacturers are not faced with such problems because they don’t actually have to demonstrate that their supplements have the health effects that they claim they have. The war against cancer goes on, but one weapon is not being developed.