Category: Acupuncture

Best Hospital Eye Roll
Science: Figuring things out is better than making things up. A tee shirt I recently saw. Except… In a recent post Mayo Clinic Promotes Reiki, Steve seemed surprised that the Mayo was offering Reiki. I don’t know. Maybe he was channeling Louie. I know the Mayo is a top hospital, but I trained in Minneapolis at Hennepin County and we would have...

More Americans turning to complementary approaches to pain control
The use of of complementary health approaches overall, and for pain management, is growing among US adults.

Boosting. What To Do.
The immune system can't be "boosted." It is an inane concept used by those who promote unscientific approaches to medicine.

Make Acupuncture Great Again
Calling a losing study a win. Making acupuncture great again.

Skeptics in the Pub. Cholera.
Prologue from the serialization of the novel Skeptics in the Pub: Cholera.
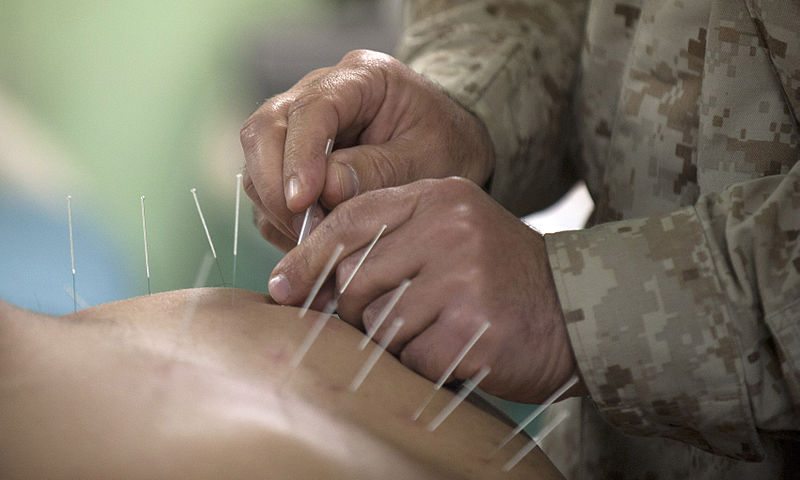
Washington Post Falls For Acupuncture Pseudoscience
An opinion piece falls for all the usual acupuncture misdirection.

Acupuncture Myths
Utilizing an evidence-based approach to evaluate acupuncture myths? Mmmmm. Not so much.
Systematic Review of Systematic Reviews of Acupuncture
No surprise - there is precious little evidence that acupuncture does anything.



