
How Unbekoming! The central delusion of MAHA
What do vitamin K shots, vaccines, MTHFR variants, and statins have in common? The answer is what I call the central delusion behind MAHA, which was inadvertently revealed by a blogger with the 'nym Unbekoming.
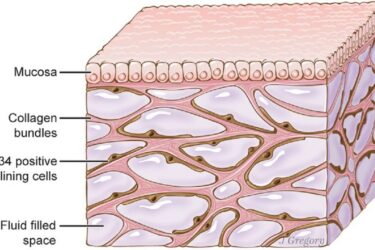
A blast from the past: The “interstitium,” the inspiration for that recent awful NYT acupuncture article
I'm on vacation this week and decided to repost a 2018 article that I had written for my other blog (but never published on SBM) that's oddly relevant to the SBM post last week about that awful NYT acupuncture article. Meet the introduction of the "interstitium" in acupuncture, complete with a major Deepak Chopra connection!

More credulous nonsense about acupuncture, this time from National Geographic
PNAS recently published credulous nonsense about acupuncture so bad that I thought it couldn't be topped. "Hold my beer!" cried National Geographic, as it proceeded to top PNAS.

MAHA vs. the FDA: Dredging up old anti-regulation revisionist history
Recently, I've noticed articles from outlets aligned with MAHA calling for the elimination of the FDA. It's all recycled "health freedom" revisionist history and ahistorical nonsense.
The myth of the magically powerful placebo returns
It's been a long time since I've written about the deceptive narratives around placebos promoted by supporters of alternative medicine. Unfortunately, a new article claiming placebos can work as well as "real medicine" is making the rounds on social media. Here we go again.
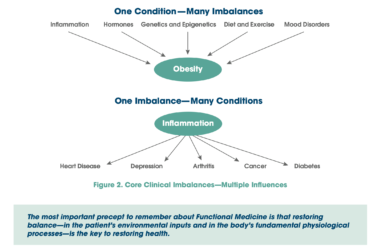
KevinMD platforms criticism of MAHA by a “functional medicine” practitioner
Although some of the criticisms of Robert F. Kennedy Jr.'s MAHA movement are spot on, why did KevinMD platform a practitioner of functional medicine woo to make them?

MAHA and science-based public health: Can’t we all just get along?
The answer is: Very likely not, at least not as long as MAHA embraces quackery and antivax pseudoscience—not that that didn't stop STAT News from ignoring the elephant in the room, vaccines, in search of a "kumbaya" moment between MAHA and public health.

Antivax tech bro Steve Kirsch uses AI to rediscover the Jock Doubleday challenge
Everything old is new again, even when AI is involved, as a very old antivax trope is rediscovered for a new generation.

RFK Jr. is definitely coming for your vaccines (part 9): ProPublica reports, and a bump in the road to remaking ACIP
A judge recently ruled that HHS Secretary Robert F. Kennedy Jr. went too far in his drive to turn ACIP into a an antivax committee. Meanwhile, ProPublica reports on where we are heading; it's not good.


