Category: Diagnostic tests & procedures
Diagnostic Reflections
At the end of a long and storied career I reflect upon making a medical diagnosis. No reflection is one way to diagnose a vampire.

TB Or Not TB? Don’t Rely on a Blood Test
TB can't be diagnosed with a blood test.
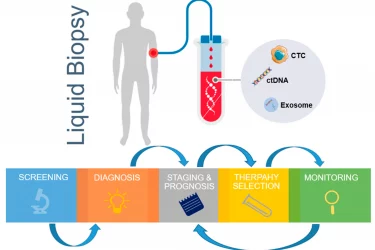
The Role of Liquid Biopsies in Oncology
Liquid biopsies can be used to reduce the amount of chemotherapy used for patients with stage II colon cancer.
Too many lab tests still escape FDA review, threatening patient safety
Even as the lab testing market grows, too many tests escape FDA review based on a meaningless categorization that has nothing to do with patient safety. Congress should pass the VALID Act of 2021, allowing the FDA to adequately regulate direct-to-consumer and other lab tests with the potential to harm the public.
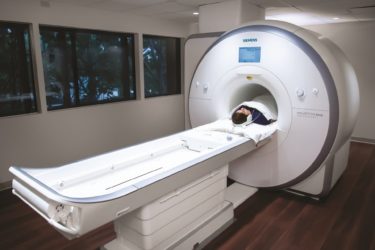
Myocarditis and how to think about it… like a cardiologist
In response to the dumpster-diving VAERS study published earlier this month, pediatric cardiologist and guest blogger Dr. Frank Han adds context by explaining how cardiologists think about and diagnose myocarditis.
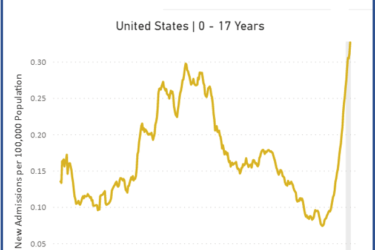
Is RSV Being Misdiagnosed as COVID-19?
Doctors who claimed that COVID-19 was essentially harmless to children are now being confronted with the sad fact that it is sometimes quite dangerous to them. How they respond reveals much about them and little about the virus.

Long COVID: Alternative medicine sees a business opportunity
With vaccines transforming the risk and consequences of a COVID infection, the health burden of "long COVID" may become a lasting consequence of the pandemic. Alternative medicine senses a business opportunity.
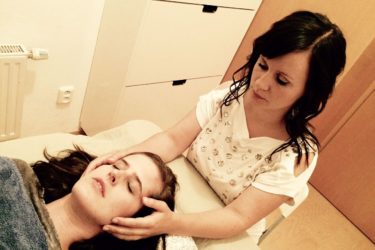
Craniosacral Therapy Is Bogus but DOs Are Required to Learn It
The standard textbook used in many schools of osteopathic medicine includes a lamentable chapter on cranial manipulation. It is clearly biased and fails to meet the minimal standards of science-based medicine. Craniosacral manipulation therapy is bogus, and it should no longer be taught to DOs or feature on their exams.
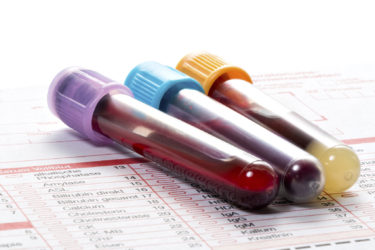
A Blood Test for Fibromyalgia?
The FM/a test is advertised as a definitive test to diagnose fibromyalgia. It isn't definitive. It is expensive. It has not been shown to change patient outcomes.
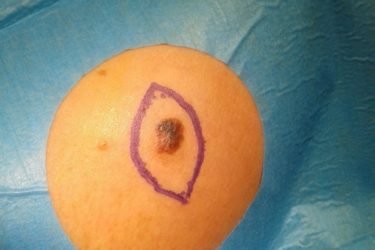
Melanoma: A Pseudoepidemic of Skin Cancer Prompts New Screening Recommendations
There appeared to be an epidemic of melanoma skin cancer, but it seems to be a pseudoepidemic caused by overdiagnosis. Screening everyone with skin exams does more harm than good and can no longer be recommended.