These do not cure cancer
One of the points I’ve tried to emphasize through my contributions to Science-Based Medicine is that every treatment decision requires an evaluation of risks and benefits. No treatment is without some sort of risk. And a decision to decline treatment has its own risks. One of the challenges that I confront regularly as a pharmacist is helping patients understand a medication’s expected long-term benefits against the risks and side effects of treatment. This dialogue is most challenging with symptomless conditions like high blood pressure, where patients face the prospect of immediate side effects against the potential for long-term benefit. One’s willingness to accept side effects is influenced, in part, by and understanding of, and belief in, the overall goals of therapy. Side effects from blood-pressure medications can be unpleasant. But weighed against the reduced risk of catastrophic events like strokes, drug therapy may be more acceptable. Willingness to accept these tradeoffs varies dramatically by disease, and are strongly influenced by patient-specific factors. In general, the more serious the illness, the greater the willingness to accept the risks of treatment.
As I’ve described before, consumers may have completely different risk perspectives when it comes to drug therapies and (so-called) complementary and alternative medicine (CAM). For some, there is a clear delineation between the two: drugs are artificial, harsh, and dangerous. Supplements, herbs and anything deemed “alternative”, however, are natural, safe, and effective. When we talk about drugs, we use scientific terms – discussing the probability of effectiveness or harm, and describing both. With CAM, no tentativeness or balance may be used. Specific treatment claims may not be backed up by any supporting evidence at all. On several occasions patients with serious medical conditions have told me that they are refusing all drug treatments, describing them as ineffective or too toxic. Many are attracted to the the simple promises of CAM, instead. Now I’m not arguing that drug treatment is always necessary for ever illness. For some conditions where lifestyle changes can obviate the need for drug treatments, declining treatment this may be a reasonable approach – it’s a kick in the pants to improve one’s lifestyle. Saying “no” may also be reasonable where the benefits from treatment are expected to be modest, yet the adverse effects from treatments are substantial. These scenarios are not uncommon in the palliative care setting. But in some circumstances, there’s a clear medical requirement for drug treatment – yet treatment is declined. This approach is particularly frustrating in situations where patients face very serious illnesses that are potentially curable. This week is the World Cancer Congress in Montreal and on Monday there were calls for patients to beware of fake cancer cures, ranging from laetrile, to coffee enemas, to juicing, and mistletoe. What are the consequences of using alternative treatments, instead of science-based care, for cancer? There are several studies and a recent publication that can help answer that question.
Who uses CAM instead of medicine?
Surveys suggest the vast majority of consumers with medical conditions use CAM in addition to, rather than as a substitute for medicine – that is, it is truly “complementary”. But there is a smaller population that uses CAM as a true “alternative” to medicine. A study by Nahin et al in 2010 looked at data from the 2002 National Healath Interview Survey (NHIS), which is described as a representative sample of Americans. It examined the group that did not use “conventional care” in the past 12 months – no health professionals, no emergency room visits, no surgery, and no nursing care of any kind. It asked about alternative medicine, which included acupuncture, ayurveda, biofeedback, chelation, chiropractic, energy healing/Reiki, hypnosis, massage, naturopathy, homeopathy, specialized diets, high-dose vitamins, yoga, tai chi, qui gong, and meditation. The survey found that 19.3% of adults did not access any “conventional” health care in the past 12 months. Of this group, over one third (38.4%) had some health need (of which 23.8% considered a serious condition). In the population that did not use conventional care, one-quarter (24.8%) used some form of alternative medicine. And 12% (approximately 4.6 million Americans) were estimated to be using alternative medicine, and not conventional medicine, to treat one or more health issues. Barriers to accessing health care were explored and users of alternative medicine had poorer health and had more barriers to care, with about 20% noting the decision to use alternative care was based on cost considerations of conventional care. There were several limitations worth noting: Most importantly, the types of conditions treated with “only CAM” were not collected. And as some users cited the costs of conventional care as a barrier, different insurance schemes might be expected to change utilization patterns. Finally, there is no assessment of outcomes. But this survey suggests that there is a small population that will preferentially treat a medical condition with CAM and not medicine.
So how would a decision to accept no treatment, or to only use alternative medicine, compare to “conventional” cancer care (chemotherapy, radiation, and surgery)? And what about delaying conventional cancer care to allow a trial of alternative medicine – does it have a measurable effect? Answering this question isn’t straightforward. In cancer research, new drugs are typically added to, or follow, established therapies, so all patients receive standard treatment options as part of their care. So we can’t ethically randomize patients to nothing, when established treatments exist. But we can answer this question in a different way: Patients that voluntarily opt out of cancer treatment can be followed, and compared to patients that do take cancer treatment. While it isn’t a prospective randomization, which would be the gold standard, it’s the best we can get. But even this approach is difficult. Most patients who decide to opt-out of cancer treatment, also opt-out of any follow-up evaluation. So tracking down patients, and their outcomes, is essential.
The effects of treatment refusals and delay, and the effectiveness of CAM as a substitute, has been evaluated in several groups of patients with breast cancer. Breast cancer is well studied, frequently diagnosed, and if detected early, potentially curable. Conventional treatment for early (localized) breast cancer is surgical resection of the tumor, followed by radiation and chemotherapy to reduce the risk of disease recurrence, by killing any residual cancer cells that remain. The overall effectiveness of conventional treatment is strongly influenced by the extent of the disease at diagnosis. When treated early in the disease course, the long-term outlook for for women with breast cancer can be excellent. However, once the cancer has spread to the lymph nodes, or metastasized to other part of the body, the outcomes are much worse. The treatment focus shifts from curative to palliative. (An old study of untreated breast cancer suggest the 5 year survival rates are 18% at 5 years and 3.6% at 10 years.) Given the potential for treatment cures, very few women elect to reject conventional treatment, or substitute CAM. But some do, which can inform us of the effectiveness of conventional care, as well as that of CAM. I found 5 studies which look at this question:
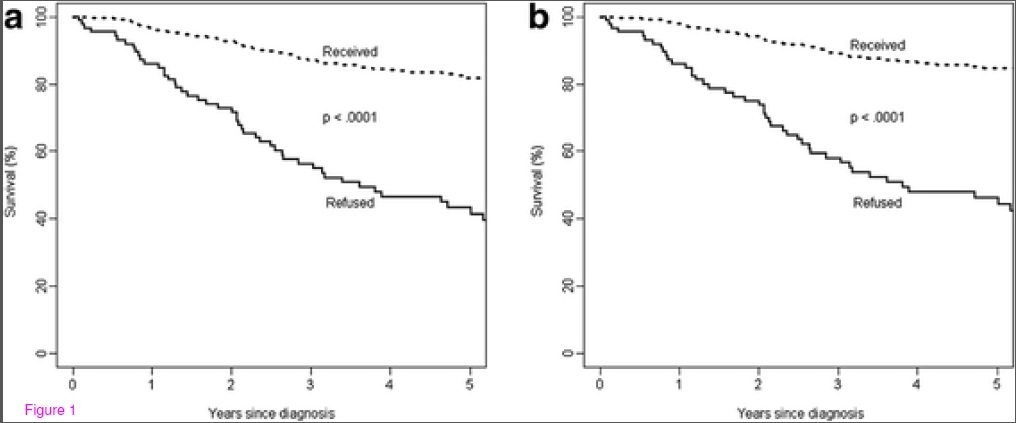
1. Patients’ Refusal of Surgery Strongly Impairs Breast Cancer Survival
This was a Swiss study by Verkooijen et al, published in 2005 in the Annals of Surgery that looked at 5339 patients under the age of 80 with non-metastatic breast cancer. It didn’t examine CAM, just the decision to refuse breast cancer surgery. It compared patients who refused breast cancer with those that those that accepted surgery. Only 1.3% of women (70) refused surgery. Of that group, 37 had no treatment, 25 had hormone-therapy only, and 8 had other types of treatments. So only a small percentage refused all treatment. In this study, the five-year survival of women that refused surgery was 72% versus 87% of women who had surgery. Adjusting for prognostic factors, the authors estimated that women that refused surgery had a 2.1-fold increased risk of death from breast cancer compared to conventional treatment. The survival curves make this clear:
The bottom line in this paper was that a decision to forgo surgery for breast cancer is associated with dramatically worse outcomes and survival.
2. Outcomes of breast cancer in patients who use alternative therapies as primary treatment
This was a medical chart review by Chang et al, published in the American Journal of Surgery in 2006. It examined breast cancer patients who refused conventional chemotherapy, or delay its initiation, in order to use CAM. The authors calculated each patient’s prognosis at the time of diagnosis. In total, 33 women were included. (Notably, patients who refused treatment and did not return for follow-up were excluded from the analysis, possibly biasing the results.) The results were grim:
- Eleven patients initially refused surgery. Ten of these patients experienced progressive disease. Five ultimately had surgery. In the six others, the cancer had already metastasized, so surgery would have offered no benefit.
- Three patients refused to allow sampling of lymph nodes to evaluate disease spread. One of these patients developed recurrent disease in the lymph nodes.
- Ten patients refused local control (surgery/radiation) of the tumor site. Two patients developed recurrences in the same location, and two developed metastatic disease.
- Nine patients refused chemotherapy, raising their estimated 10-year mortality from 17% to 25%
Consistent with the study above, the vast majority of breast cancer patients who refuse surgical intervention developed progressive disease. Even delaying surgery increased risks and overall mortality. Outcomes were better for patients that accepted surgery, but refused adjuvant treatments, like chemotherapy. However, even this strategy significantly raised 10-year mortality estimates.
3. Alternative therapy used as primary treatment for breast cancer negatively impacts outcomes
This study from Han et al was published in the Annals of Surgical Oncology in 2011, and may include some of the patients in the Chang analysis. This was a retrospective chart review of breast cancer patients who refused or delayed conventional treatments. 61 patients were identified. On average patients had Stage 2 disease at diagnosis, which is highly treatable and potentially curable. In patients that omitted or delayed surgery (26 women) 96.2% had disease progression with 50% dying of the disease. At the time of diagnosis, the median tumor size was 2.0cm. Upon follow-up, the median-size was 7.8cm. (The authors include this photo of a tumor in a woman who elected to use diet and herbal treatments, rather than receive surgery.)
In patients that accepted surgery but rejected adjuvant therapy (chemotherapy/radiation), the initial 10-year relapse-free survival was estimated at 59.2%, which would have been 74.3% had the patients accepted these treatments. Actual outcomes were much worse than predicted. The actual observed relapse-free survival was only 13.8%. Some subsequently elected to take palliative chemotherapy and radiation to control their disease. From this study we can conclude that refusing or delaying conventional cancer care is associated with much worse outcomes.
4. Prognosis following the use of complementary and alternative medicine in women diagnosed with breast cancer
This analysis, by Saquib et al, was a secondary analysis of the Women’s Healthy Eating and Living (WHEL) study. It looked at 2562 breast cancer survivors and surveyed for rejection of systemic treatment (i.e., chemotherapy) and use of CAM following surgical resection. All women had to be aged 18-70 and had operable Stage I-IIIa breast cancer. In this group, 177 women were identified who declined systemic treatment. 80% of this group used CAM. Compared to women that took chemotherapy, women that declined systemic treatment had a 90% greater risk of an additional breast cancer event, and the risk of death increased by 70%. CAM use had no effect on this finding. In addition, the lack of effect was consistent between “high supplement users” (>3 per day) and low supplement users. The authors concluded that women that decline systemic treatment are at greater risk for subsequent recurrence and death due to breast cancer. The use of CAM had no measurable effect on the recurrence of breast cancer or on the risk of subsequent death.
5. Outcome analysis of breast cancer patients who declined evidence-based treatment
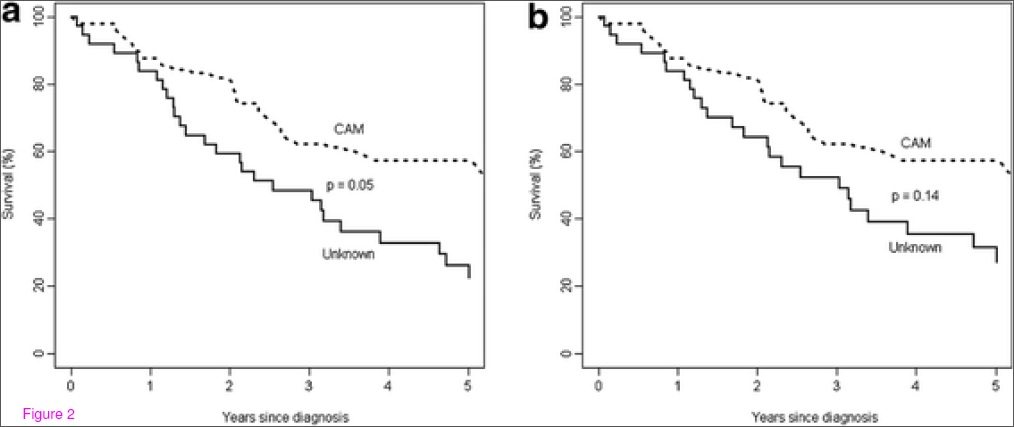
Here is the recent paper I referred to above, which studied women with breast cancer in Northern Alberta who refused standard treatments. It was also a chart review with a matched pair analysis (age, disease stage, calendar year) that compared survival with those that received conventional cancer care. Between 1980 and 2006 they identified 185 women (1.2%) that refused cancer care following diagnosis by biopsy. (Notably, cancer care is an insured service in Alberta, so there should have been no financial barriers in accessing treatment.) Women older than 75 were excluded from the analysis because this population is generally not included in clinical trials and active treatment regimens. In addition, women that accepted surgery, but rejected chemotherapy/radiation were excluded from the analysis. To qualify, women had to have rejected all conventional care. The final population studied was 87 women, most of whom presented with early (Stage I or II) disease. Most were married, over the age of 50, and urban residents. In this group, the primary treatment was CAM in 58%, and was unknown in the remainder. Some women in this group eventually accepted cancer care, and the average delay was 20-30 weeks due to CAM use.
The results were grim. The 5 year overall survival was 43% for women that declined cancer care, and 86% for women that received conventional cancer care. For cancer-specific survival (i.e., those that died of breast cancer) survival was 46% vs. 85% in those that took cancer care. The survival curves are ugly:
The authors compared the “CAM” group to those where treatment plan (if any) was not known:
Note that the difference is only statistically significant in (a) where all-causes of death were included and not (b), cancer-specific causes. So does that mean CAM helps? Probably not. The two groups are not well defined, and the “unknown” group could include CAM users – it is not a comparison of CAM versus no treatment. And as the types of CAM used was not documented, this is a heterogeneous group. The key point this comparison illustrates is that CAM users did dramatically worse than women that took conventional cancer care. Even delaying surgery to allow for CAM first significantly decreased the effectiveness of subsequent conventional care. The authors conclude, correctly, that there is no evidence to support using CAM as primary cancer treatment.
Effectiveness evaluations
The data show that avoiding or delaying conventional cancer care is associated with negative outcomes, and CAM used does not seem to modify this risk. But have any specific CAM interventions shown any benefit? Probably the most comprehensive single review is a systematic review by Gerber et al, published in Breast Cancer Research and Treatment in 2006, which looked at CAM effectiveness for early breast cancer. It concludes:
There is no compelling evidence that any of the numerous complementary treatments available is sufficiently effective in breast cancer patients to justify its use. It should be the responsibility of those who claim efficacy for CAM to support these claims with acceptable evidence, rather than the responsibility of those who criticize CAM to prove its non-efficacy.
Based on the current evidence, there is nothing to suggest that any specific CAM treatment has any meaningful clinical effects.
Conclusion: Alternative medicine isn’t real medicine
Despite widespread claims, there is no evidence to support the use of any CAM treatment as a replacement for conventional cancer care. As the studies in breast cancer show, delaying treatment or substituting CAM for conventional cancer care dramatically worsens outcomes.The results of these studies will hopefully provide patients and health providers with a better understanding of the risks and consequences of CAM for cancer. CAM is no alternative to science-based cancer care.
References
1. Verkooijen HM, Fioretta GM, Rapiti E, Bonnefoi H, Vlastos G, Kurtz J, Schaefer P, Sappino AP, Schubert H, & Bouchardy C (2005). Patients’ refusal of surgery strongly impairs breast cancer survival. Annals of surgery, 242 (2), 276-80 PMID: 16041219
2. Chang EY, Glissmeyer M, Tonnes S, Hudson T, & Johnson N (2006). Outcomes of breast cancer in patients who use alternative therapies as primary treatment. American journal of surgery, 192 (4), 471-3 PMID: 16978951
3. Han E, Johnson N, DelaMelena T, Glissmeyer M, & Steinbock K (2011). Alternative therapy used as primary treatment for breast cancer negatively impacts outcomes. Annals of surgical oncology, 18 (4), 912-6 PMID: 21225354
4. Saquib J, Parker BA, Natarajan L, Madlensky L, Saquib N, Patterson RE, Newman VA, & Pierce JP (2012). Prognosis following the use of complementary and alternative medicine in women diagnosed with breast cancer. Complementary therapies in medicine, 20 (5), 283-90 PMID: 22863642
5. Joseph K, Vrouwe S, Kamruzzaman A, Balbaid A, Fenton D, Berendt R, Yu E, & Tai P (2012). Outcome analysis of breast cancer patients who declined evidence-based treatment. World journal of surgical oncology, 10 (1) PMID: 22734852