Tag: right to try laws
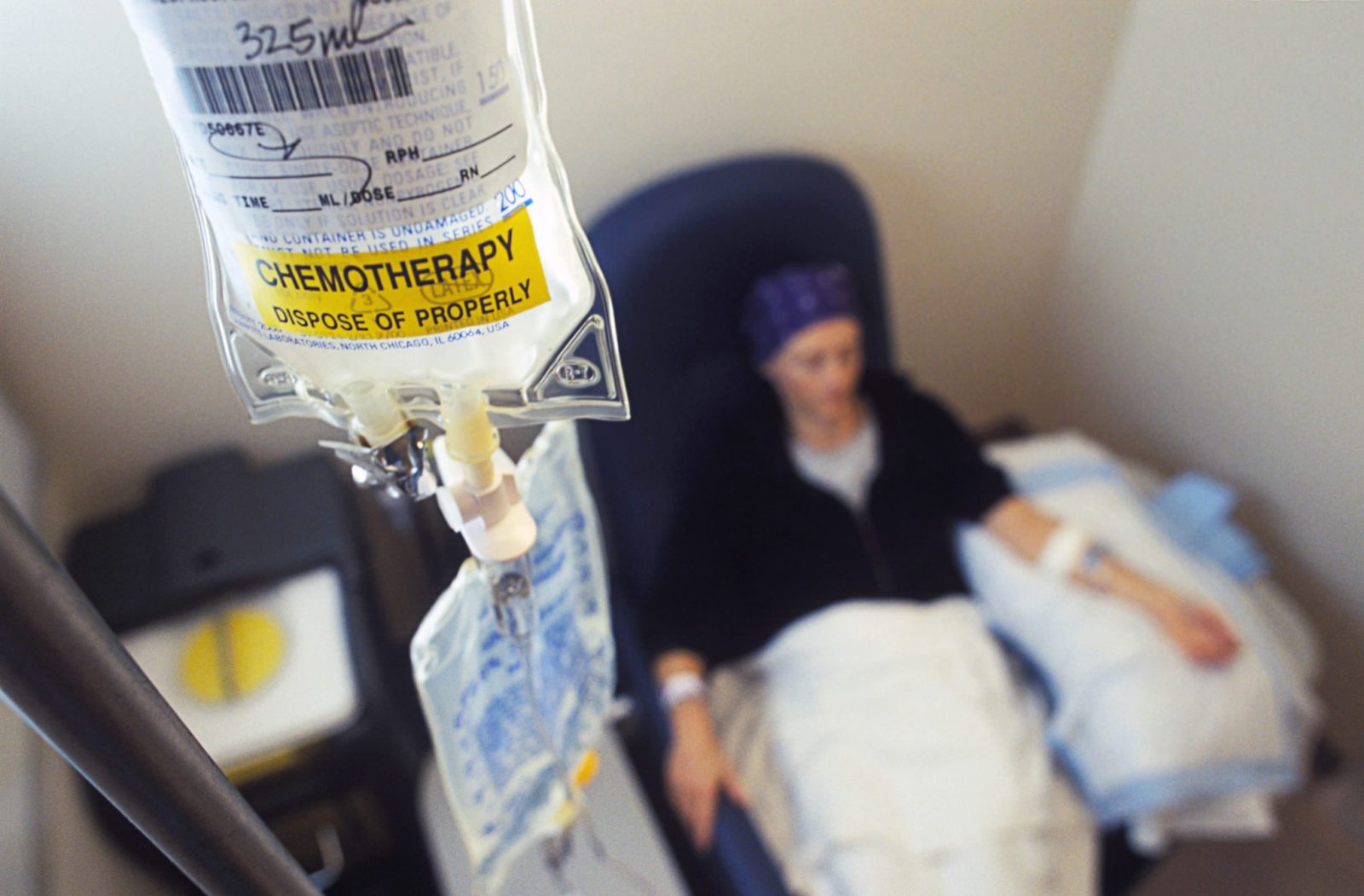
Bills remove impediments to ill-advised state “right to try” laws, shield wrongdoers, and hide adverse events
Congressional bills will unleash state "right to try" laws, block terminally ill patients from redress for damages caused by negligent doctors and drug companies, and hide adverse drug events from the public.

FDA Looks At Dubious Stem Cell Clinics
Using stem cells to treat disease or improve recovery is an exciting area of research. The potential is undeniably great – these are cells that have the potential to differentiate into mature cells of a specific type. They can be used to replace damaged cells or improve the environment for cell function and recovery. Ideally stem cells can be developed from cells...

As in 2014, “right-to-try” laws continue to metastasize in 2015, part 2
"Right-to-try" laws sound on the surface to be reasonable and compassionate. They are neither. And they continue to metastasize from state to state.

As in 2014, “right-to-try” laws continue to metastasize in 2015
Last year, I did several posts on what I consider to be a profoundly misguided and potentially harmful type of law known as “right-to-try.” Beginning about a year and a half ago, promoted by the libertarian think tank known as the Goldwater Institute, right-to-try laws began popping up in state legislatures, which I likened to Dallas Buyers Club laws. Both Jann Bellamy...

Using the fear of Ebola to promote the placebo legislation that is “right to try”
Libertarians and free market fundamentalists generally detest the FDA and want to dramatically decrease its power in the belief that the free market can guarantee the safety of drugs better than a government agency that requires strong scientific evidence of efficacy and safety before approval. Not surprisingly, they're at it again, this time in the service of promoting "right-to-try" laws and using...
“Right to try” laws and Dallas Buyers’ Club: Great movie, terrible for patients and terrible policy
One of my favorite shows right now is True Detective, an HBO show in which two cops pursue a serial killer over the course of over 17 years. Starring Woody Harrelson and Matthew McConaughey, it’s an amazingly creepy show, and McConaughey is amazing at playing his character, Rustin Cohle. I’m sad that the show will be ending tomorrow, but I really do...
The illusions of “right to try” laws
[Ed. Note: For additional commentary on why “right-to-try” laws are such a bad idea, see “Right to try” laws and Dallas Buyers’ Club: Great movie, terrible for patients and terrible policy and The false hope of “right-to-try” metastasizes to Michigan.] There is nothing like a touching anecdote to spur a politician into action. And those who want to try investigational drugs outside...


